Inflammation is how our cells respond to stress, but too much can be dangerous. To control this response, researchers studied enzymes that act like switches. They focused on p38α, a key regulator in inflammation. The research shows that p38α is turned on by another enzyme in a precise dance-like interaction. Understanding this could help stop dangerous inflammation and study other diseases like cancer and Alzheimer’s.
Cells face stress, like infections, which can disrupt normal body functions. To cope with stress, cells use inflammation, but too much can be harmful, causing severe illnesses like cytokine storms, as seen in COVID-19. EMBL Grenoble and University of Geneva researchers studied a protein called p38α, which acts as a switch for inflammation. They figured out how p38α is turned on by another protein called MKK6. This discovery could help make drugs to control cytokine storms and improve our understanding of diseases.
Inflammation is like a series of switches turning on, with each switch activating the next one. The final button, called p38α, triggers genes for inflammation. Too much inflammation can cause dangerous cytokine storms. Researchers want to stop this last switch because it’s an important drug target.
Researchers have studied p38α, but there are still missing pieces in the puzzle. Most drugs try to stop p38α by targeting a standard part shared by many kinases. However, this lacks specificity and can affect other processes. So far, no drugs have been successful in clinical trials for this reason.
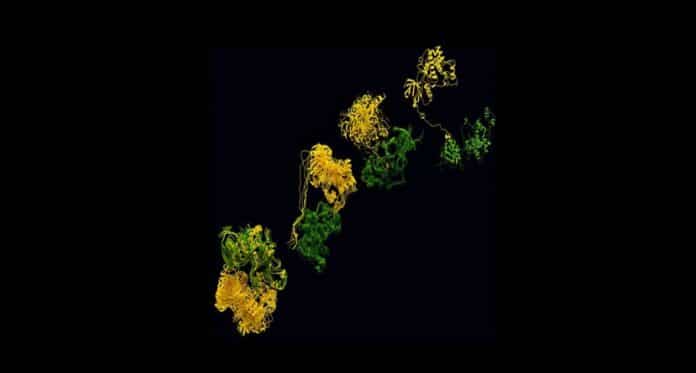
By using advanced techniques like cryo-EM and SAXS, the research team successfully captured the 3D structure of the complex. They also discovered a previously unknown docking site where the two enzymes interact. This newfound knowledge is crucial for understanding the activation process of p38α.
Pauline Juyoux, a former researcher at EMBL Grenoble the first author of the study, said, “This could be an interesting target for inhibitors to block this specific interaction, and therefore the signal triggering the inflammatory response.”
The Gervasio Lab at University of Geneva uncovered how the two kinases join together after a dynamic ‘dance,’ starting with a ‘hug’ of the KIM domain and ending with a face-to-face dimer formation. Using advanced simulations alongside experimental data like SAXS and Cryo-EM, they could visualize the interaction between the kinases, almost like a ‘courtship’ dance. This discovery provides a new target for potential drugs. It could also help understand similar processes in other kinase families linked to cancer and Alzheimer’s.
In conclusion, understanding the “dance” of signaling proteins like p38α represents a significant step towards unraveling the complexities of inflammation and potentially developing more targeted and effective therapies to manage this crucial biological response.
Journal reference:
- Pauline Juyoux, Ioannis Galdadas, et al., Architecture of the MKK6-p38α complex defines the basis of MAPK specificity and activation. Biorxiv. DOI: 10.1101/2022.07.04.498667.