Obesity has become a global health crisis, leading to an increase in conditions like type 2 diabetes and nonalcoholic steatohepatitis. Researchers from the University of California San Diego School of Medicine have uncovered new insights into how obesity impacts mitochondria, the essential energy-producing structures in our cells.
In a new study, the researchers observed that mice fed a high-fat diet experienced a breakdown of mitochondria within their fat cells. These mitochondria fragmented into smaller units with reduced capacity for burning fat. The researchers identified a single gene that controls this process. Deleting this gene in the mice protected them from excessive weight gain, even when consuming the same high-fat diet as other mice.
Alan Saltiel, Ph.D., professor in the Department of Medicine at UC San Diego School of Medicine, said, “Caloric overload from overeating can lead to weight gain and also triggers a metabolic cascade that reduces energy burning, making obesity even worse. The gene we identified is a critical part of that transition from healthy weight to obesity.”
In situations of caloric imbalances, such as in obesity, the efficiency of fat cells in burning energy diminishes. This difficulty in burning energy contributes to the challenge people with obesity face in losing weight. Understanding how these metabolic abnormalities begin is a major puzzle in the study of obesity.
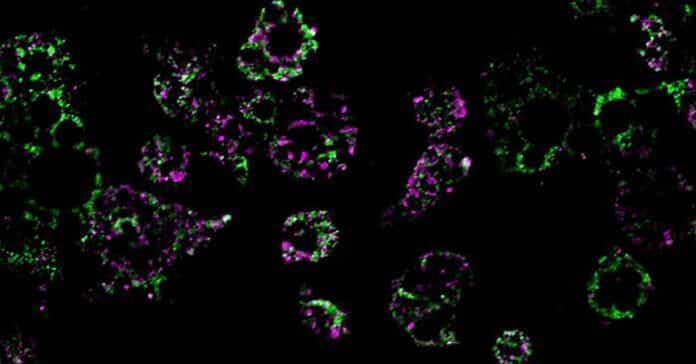
To investigate this, researchers fed mice a high-fat diet and observed its impact on the mitochondria of their fat cells—the cellular structures responsible for burning fat. They made a noteworthy discovery: after exposure to a high-fat diet, the mitochondria in certain parts of the mice’s adipose tissue underwent fragmentation. This resulted in the mitochondria splitting into numerous smaller and less effective units that burned less fat.
Furthermore, the researchers identified a key player in this metabolic process: a single molecule called RaIA. RaIA serves various functions, including breaking down malfunctioning mitochondria. The study suggests that when RaIA is overly active, it disrupts the normal functioning of mitochondria, triggering the metabolic issues associated with obesity. This finding sheds light on a potential target for addressing the root causes of metabolic abnormalities in obesity.
Saltiel said, “In essence, chronic activation of RaIA appears to play a critical role in suppressing energy expenditure in obese adipose tissue. By understanding this mechanism, we’re one step closer to developing targeted therapies that could address weight gain and associated metabolic dysfunctions by increasing fat burning.”
By deleting the gene associated with RaIA, the researchers protected the mice against weight gain caused by a high-fat diet. Upon further investigation into the biochemical processes involved, the researchers identified that some proteins influenced by RaIA in mice have human counterparts associated with obesity and insulin resistance. This suggests that similar mechanisms may be at play in driving obesity in humans.
Saltiel said, “The direct comparison between the fundamental biology we’ve discovered and real clinical outcomes underscores the relevance of the findings to humans and suggests we may be able to help treat or prevent obesity by targeting the RaIA pathway with new therapies. We’re only beginning to understand this disease’s complex metabolism, but the future possibilities are exciting.”
Journal Reference:
- Xia, W., Veeragandham, P., Cao, Y. et al. Obesity causes mitochondrial fragmentation and dysfunction in white adipocytes due to RalA activation. Nat Metab (2024). DOI: 10.1038/s42255-024-00978-0