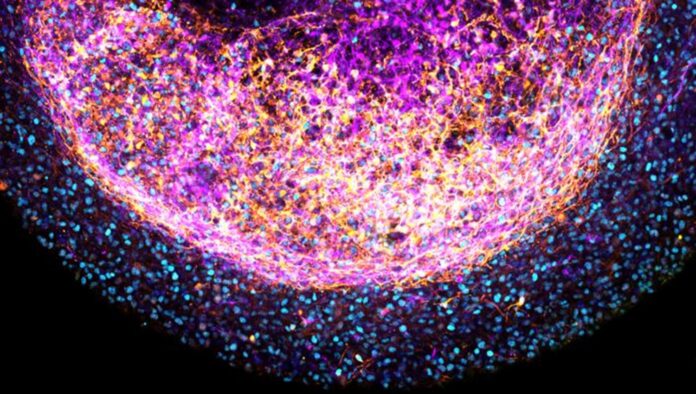
Scientists in labs create models of healthy tissues and diseases in various ways. One recent method involves using 3D mini-organs, known as organoids, to mimic the functions of an organ closely. These organoids can be generated directly from cells of a tissue or by guiding stem cells to develop into the desired organ.
Brain organoids, for instance, were traditionally grown by encouraging stem cells to form structures resembling different regions of the brain, using specific chemical combinations to mimic natural brain development.
Scientists at the Princess Máxima Center for Pediatric Oncology and the Hubrecht Institute, based in Utrecht, the Netherlands, developed brain organoids directly from human fetal brain tissue. These organoids can self-organize in vitro.
Scientists made a surprising discovery in growing mini-brains. Instead of breaking down the original fetal brain tissue into individual cells, they found that using small pieces of the tissue allowed it to self-organize into complex organoids resembling the human brain.
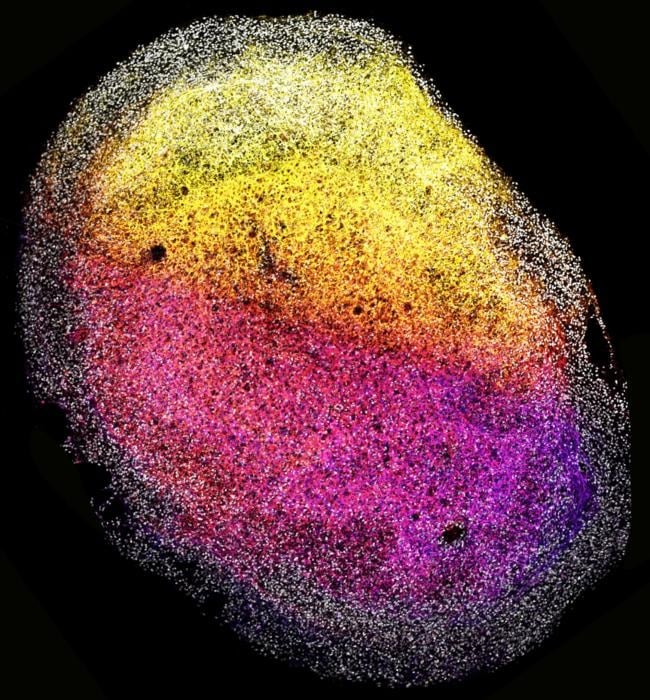
These brain organoids, about the size of a grain of rice, contained various brain cells and were particularly rich in outer radial glia, a cell type found in humans and our evolutionary ancestors. This finding enhances the similarity of these organoids to the human brain and their utility in studying brain functions.
Using whole pieces of fetal brain tissue to grow mini-brains produced extracellular matrix proteins, forming a supportive structure around cells. This unique feature allowed the brain tissue to self-organize into complex 3D systems resembling the human brain.
The presence of extracellular matrix in the organoids provides an opportunity to study the brain cell environment and understand potential dysregulations. These tissue-derived organoids retained characteristics of the specific brain region they originated from, responding to signaling molecules crucial in brain development.
This discovery holds promise for unraveling the intricate network of molecules guiding brain development.
Dr. Benedetta Artegiani, research group leader at the Princess Máxima Center for Pediatric Oncology who co-led the research, says: “Our new, tissue-derived brain model allows us to better understand how the developing brain regulates the identity of cells. It could also help understand how mistakes in that process can lead to neurodevelopmental diseases such as microcephaly, as well as other diseases that can stem from derailed development, including childhood brain cancer.”
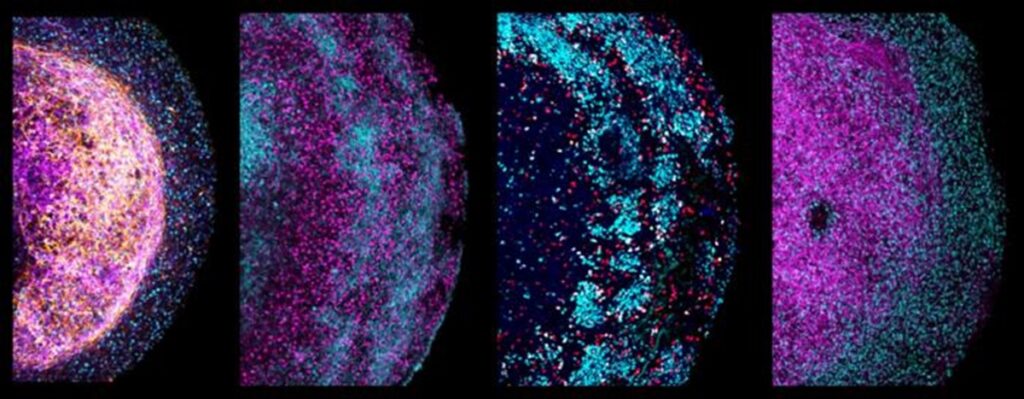
The scientists explored the potential of tissue-derived organoids in modeling brain cancer by using the CRISPR-Cas9 gene-editing technique to introduce faults in the TP53 cancer gene. Within three months, the cells with defective TP53 had outpaced healthy cells in the organoids, exhibiting a growth advantage characteristic of cancer cells.
Additionally, the team disabled three genes associated with glioblastoma, a brain tumor, using CRISPR-Cas9. These mutated organoids served as a platform for studying the response to existing cancer drugs, highlighting their potential in cancer drug research and linking drugs to specific gene mutations.
The tissue-derived brain organoids, derived from small pieces of fetal brain tissue, demonstrated the ability to self-organize into 3D brain structures. They maintained characteristics specific to the brain region from which they originated and responded to signaling molecules relevant to brain development.
The organoids were utilized to model brain cancer by introducing faults in the TP53 cancer gene using CRISPR-Cas9. These mutated organoids demonstrated sustained growth for over six months, and their ability to multiply enhances the reliability of experiments, providing a promising platform for cancer drug research and further exploration of brain development.
Future research will delve into tissue-derived brain organoids’ potential and ethical considerations.
Dr. Delilah Hendriks, affiliated group leader at the Princess Máxima Center for Pediatric Oncology, postdoctoral researcher at the Hubrecht Institute, and Oncode Investigator who co-led the research, says:
“With our study, we’re making an important contribution to the organoid and brain research. Since we developed the first human gut organoids in 2011, it’s been great to see that the technology has really taken off. Organoids have since been developed for almost all tissues in the human body, both healthy and diseased – including an increasing number of childhood tumors.”
“Until now, we were able to derive organoids from most human organs, but not from the brain – it’s really exciting that we’ve now been able to jump that hurdle as well.”
Journal Reference:
- Delilah Hendriks, Anna Pagliaro, Francesco Andreatta et al. Human fetal brain self-organizes into long-term expanding organoids. Cell. DOI: 10.1016/j.cell.2023.12.012