Mimicking natural evolution, directed evolution guides the synthesis of proteins with improved or new functions. The technique is powerful enough that it can engineer proteins.
In this technique, the original protein is mutated to create a collection of mutant protein variants. The protein variants then show improved or more desirable functions selected. These selected proteins are then afresh changed to make another accumulation of protein variations for another round of selection.
This cycle is repeated until a last, transformed protein is evolved with optimized performance contrasted with the original protein.
Now, scientists at the EPFL have used this powerful technique to create synthetic nanoparticles as optical biosensors-tiny devices that use light to detect biological molecules in air, water, or blood.
Scientists used directed evolution to modify the optoelectronic properties of DNA-wrapped single-walled carbon nanotubes (or, DNA-SWCNTs, as they are abbreviated), which are nano-sized tubes of carbon atoms that resemble rolled-up sheets of graphene covered by DNA. When they detect their target, the DNA-SWCNTs emit an optical signal that can penetrate through complex biological fluids, like blood or urine.
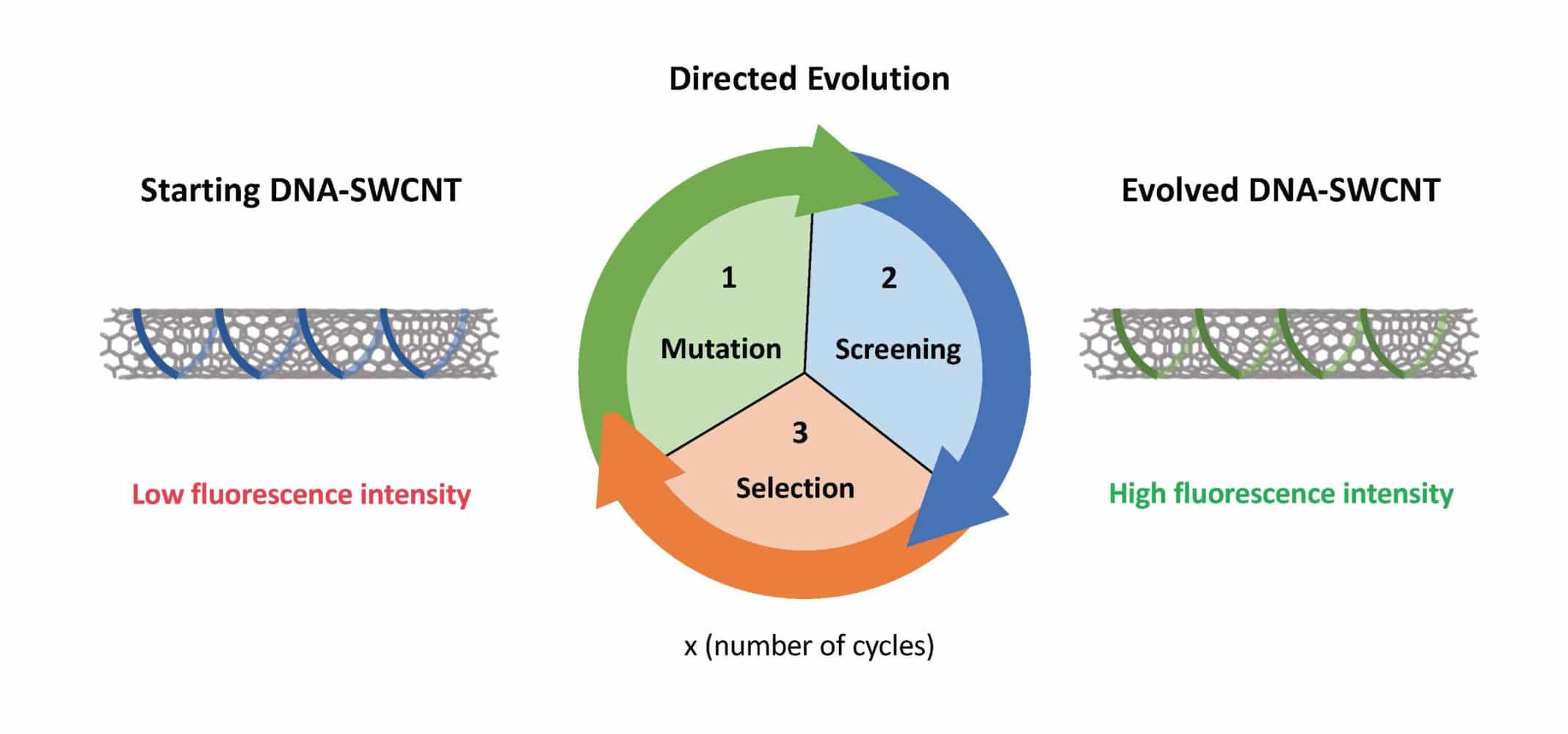
Using the technique allowed scientists to engineer new DNA-SWCNTs with optical signals that are increased by up to 56%.
Ardemis Boghossian at EPFL, said, “The majority of researchers in this field just screen large libraries of different materials in hopes of finding one with the properties they are looking for. In optical nanosensors, we try to improve properties like selectivity, brightness, and sensitivity. By applying directed evolution, we provide researchers with a guided approach to engineering these nanosensors.”
“The study shows that what is essentially a bioengineering technique can be used to more rationally tune the optoelectronic properties of certain nanomaterials.”
“Fields like materials science and physics are mostly preoccupied with defining material structure-function relationships, making materials that lack this information difficult to engineer. But this is a problem that nature solved billions of years ago – and, in recent decades, biologists have tackled it as well. I think our study shows that as materials scientists and physicists, we can still learn a few pragmatic lessons from biologists.”
The study is published in the journal Chemical Communications.