Diseases like cancer and degenerative conditions often happen because the cells don’t interact with each other appropriately. There are tiny pathways inside the cells, like roads in a city, that help them grow and function. These pathways are called “microtubules,” like microscopic protein filaments.
Researchers from the University of Geneva have found a new way these microtubules grow and function. This discovery could lead to new treatments for diseases that affect our cells. Their findings were published in the Proceedings of the National Academy of Sciences (PNAS).
Like a city needs good transportation networks for growth, cells need these microtubule “roads” to develop. When these roads aren’t working right, it can lead to diseases like cancer and neurodegenerative disorders.
It is important to figure out how these microtubules work and how they can control their growth. Even though researchers have made progress in this area, it’s still a complex puzzle that needs more research.
There are two important proteins called CLIP-170 and EB3. A researcher named Charlotte Aumeier at the UNIGE Faculty of Science found something interesting about these proteins and how microtubules work. She discovered that these two proteins, CLIP-170 and EB3, act like oil droplets in water. They separate from the watery part of the cell and come together at the tip of the microtubule as a particular liquid phase. This helps the microtubule grow.
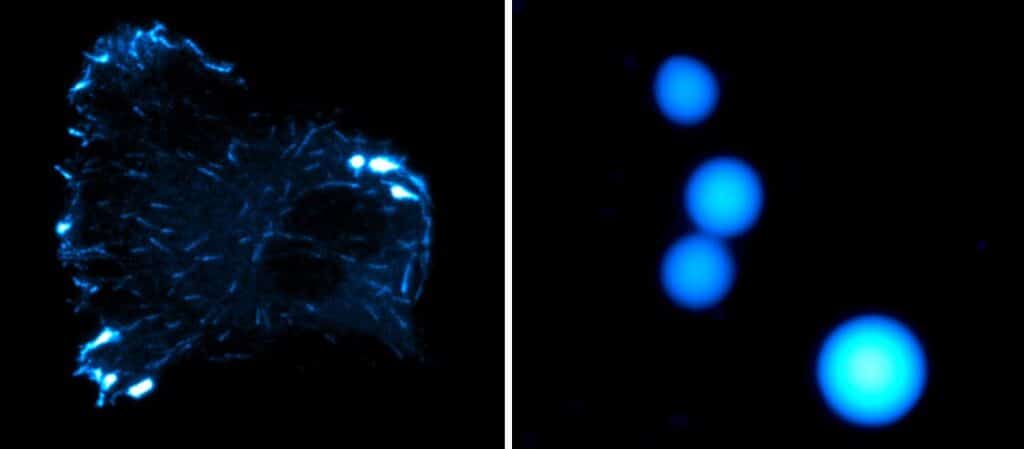
Charlotte Aumeier, the study’s last author, explains, “Microtubules are dynamic structures that continually build and deconstruct themselves. This phenomenon of phase separation, at the microtubule level, increases the concentration of proteins, including tubulin, and significantly stimulates the growth rate of microtubules while reducing depolymerisation events, i.e., microtubule decay events.” This mechanism directly and physically impacts how cellular microtubules behave.
Julie Miesch, a Ph.D. student, and her team found that two proteins, CLIP-170 and EB3, work together to control the growth of microtubules in cells. They do this through a special liquid phase separation process. When these proteins are alone, they don’t interact with the microtubules properly. But they can control how fast the microtubules grow in specific areas when they team up.
They used advanced microscopy techniques at the UNIGE ACCESS GENEVA platform to understand this. These findings show a new way to control microtubules, which could lead to new cancer treatments. It also helps us know cells better and how to intervene in their processes.
This study helps to understand how things work inside the cells, especially when it comes to tiny microtubules. It shows that two proteins, CLIP-170 and EB3, work together like a team to control how microtubules grow. They do this by separating from the cell’s liquid and forming their special liquid area.
This discovery is important because it can help to learn more about how cells function. It could lead to new ways to treat different diseases.
Journal Reference:
- Julie Miesch, Robert T. Wimbish et al., Phase separation of +TIP networks regulates microtubule dynamics. Proceedings of the National Academy of Sciences. DOI: 10.1073/pnas.2301457120.