The strengths of uniform microstructures are frequently emphasized in studies on mechanical size effects in nanosized metals, emphasizing the importance of intrinsic microstructures and extrinsic dimensions for understanding size-dependent features.
In a new study, Caltech scientists developed a hydrogel infusion-based additive manufacturing (AM) technique for printing objects a thousand times smaller: 150 nanometers, comparable to a flu virus size. This technique is similar to previously developed fabrication techniques for printing thick and microsized metal parts. Each step has been reimagined to work at the nanoscale in this new technique.
Making a photosensitive “cocktail” is the first step in the procedure. This “cocktail” is mainly made of hydrogel, a type of polymer that can absorb its weight in water many times. Then, using a laser, this mixture is selectively hardened to create a 3D scaffold that is the same shape as the required metal products. The objects in this study were nanolattices and a collection of tiny pillars.
The hydrogel parts are then injected with a nickel-ion-containing aqueous solution. The components are then baked until the hydrogel is completely burned off, leaving parts that are precisely the same shape as the original but smaller and made entirely of metal ions that have been oxidized (attached to oxygen atoms). The oxygen atoms are chemically removed from the components in the last stage, returning the metal oxide to a metallic state.

The parts develop their unexpected strength in the final step.
Julia R. Greer, the Ruben F. and Donna Mettler Professor of Materials Science, Mechanics, and Medical Engineering, said, “There are all these thermal and kinetic processes occurring simultaneously during this process, and they lead to a very, very messy microstructure. You see defects like pores and irregularities in the atomic structure, typically considered strength-deteriorating defects. If you were to build something out of steel, an engine block, you would not want to see this microstructure because it would significantly weaken the material.”
“However, they found exactly the opposite. The many defects that would weaken a metal part at a larger scale strengthen the nanoscale parts instead.”
When a pillar is free of flaws, failure happens catastrophically along what is known as a grain boundary or the location where the material’s small crystals rub up against one another.
However, a failure cannot simply spread from one grain boundary to the next when the material is packed with flaws. As a result, the distortion becomes more uniformly distributed throughout the material, indicating that the material won’t suddenly fail.
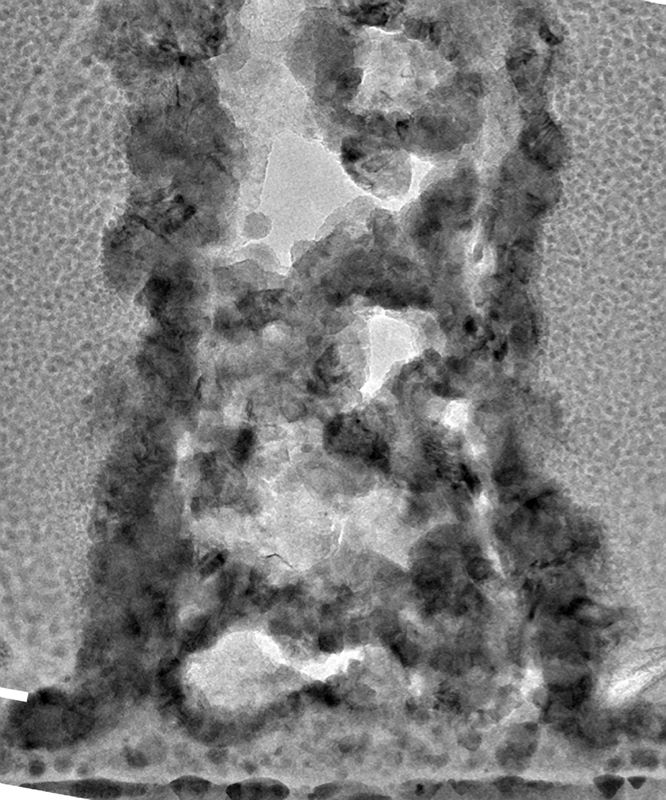
Wenxin Zhang, lead author of the work and a graduate student in mechanical engineering, said, “Usually, the deformation carrier in metal nanopillars—that is, a dislocation or slip—propagates until it can escape at the outer surface. But in the presence of interior pores, the propagation will quickly terminate at the surface of a pore instead of continuing all the way through the entire pillar. As a rule of thumb, it’s harder to nucleate a deformation carrier than to let it propagate, explaining why the present pillars may be stronger than their counterparts.”
This is one of the first demonstrations of 3-D printing of metal structures at the nanoscale. This technique is expected to create many useful components, such as catalysts for hydrogen, storage electrodes for carbon-free ammonia and other chemicals, and essential parts of devices such as sensors, microrobots, and heat exchangers.
Journal Reference:
- Wenxin Zhang, Zhi Li, Ruoqi Dang, et al. Creating many useful components, such as catalysts for hydrogen, storage electrodes for carbon-free ammonia and other chemicals, and essential parts of devices such as sensors, microrobots, and heat exchangers. Nano Letters. DOI: 10.1021/acs.nanolett.3c02309
