A team of UCL and Imperial College London researchers explored the importance of a specific brain protein called ACBD6. This protein, which regulates certain activities, was not well understood before. The study, recently published in Brain, found that ACBD6 is crucial for a healthy nervous system.
The research revealed that when ACBD6 doesn’t function properly, it leads to a rare condition in children called Autosomal Recessive ACBD6-related disorder. This condition is identified by developmental delays in thinking and movement skills, along with symptoms like dystonia and parkinsonism.
The breakthrough in understanding this disorder was achieved through advanced genomic technologies and collaboration among 89 clinicians and scientists from 72 institutes worldwide who shared extensive data.
The study of a rare disorder started with three siblings in one family who had a complex neurological condition due to mutations in the ACBD6 gene. More families with similar genetic issues were discovered through years of global collaboration. As researchers compared those affected’s clinical and radiological features, commonalities emerged.
The team examined 45 individuals from 28 families and even studied animal models. Led by Professor Ed Tate from Imperial’s Chemistry Department, the researchers specifically examined how ACBD6 deficiency affects N-myristoylation, a type of protein modification.
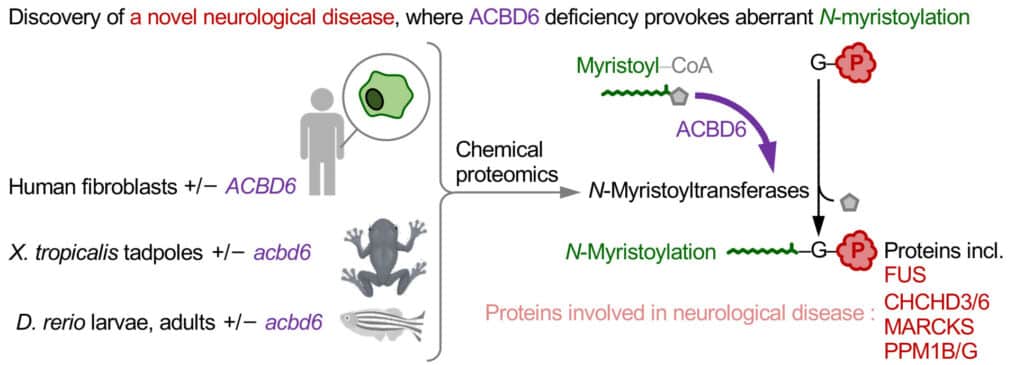
Dr Wouter Kallemeijn, from The Francis Crick Institute and the Department of Chemistry at Imperial, said: “We were able to corroborate our patient findings in ACBD6-deficient frog and zebrafish models – which suffer from remarkably similar disease symptoms as human ACBD6-deficient patients – indicating ACBD6 plays a significant role in N-myristoylation, including that of proteins crucial for and during neurodevelopment.”
Dr. Reza Maroofian, a co-lead author from the UCL Institute of Neurology, emphasized the significance of studying many individuals with ultra-rare disorders. He highlighted the valuable insights into human biology and pathology that such studies can provide, stressing the need to recognize and fund these often overlooked and underfunded research efforts. The international collaboration showcased the dedication and expertise of the global scientific community, highlighting the critical importance of not neglecting ultra-rare conditions.
Dr. Rauan Kaiyrzhanov, another co-lead author from the UCL Institute of Neurology, explained that “the immediate impact of the study is introducing these genetic disorders to the medical community, aiding in the global diagnosis of affected families. In the long run, the study contributes to a better understanding of ultra-rare conditions characterized by abnormal movements like dystonia and parkinsonism.”
This understanding, in turn, can advance our knowledge of the biological mechanisms underlying more common neurodegenerative movement disorders, such as Parkinson’s disease and dystonia.
This study represents a significant leap forward in uncovering the molecular causes of a rare neurological condition in children. By identifying the role of ACBD6 and the implications of its defects, the research contributes not only to immediate clinical applications but also to the broader field of neurodegenerative disorders.
The international collaboration underscores the importance of collective efforts in advancing scientific understanding and addressing neglected ultra-rare conditions.
Journal reference:
- Rauan Kaiyrzhanov, Aboulfazl Rad, et al., Bi-allelic ACBD6 variants lead to a neurodevelopmental syndrome with progressive and complex movement disorders. Brain. DOI: 10.1093/brain/awad380.