Major depressive disorder (MDD) significantly contributes to the global disease burden and is a leading cause of disability worldwide. Insight into disease pathophysiology and novel therapeutics are urgently needed, as treatment resistance is common and occurs in up to 30% of MDD patients.
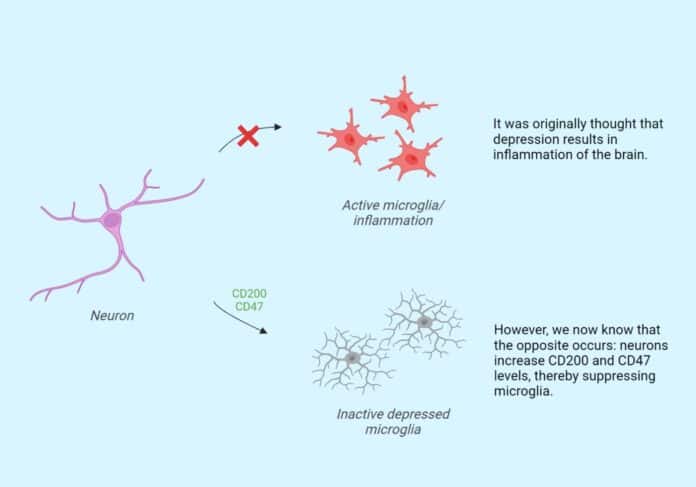
Previous studies demonstrated altered inflammatory marker levels in depressed patients. Additionally, chronic inflammatory conditions like rheumatism, inflammatory bowel disease, and multiple sclerosis have all been associated with depression. These findings imply that brain inflammation may contribute to depression. But is it the case?
A new study by Karel Scheepstra and his team analyzed post-mortem human brain tissue from people with depression. This brain tissue was contributed to the Dutch Brain Bank for Psychiatry (NHB-Psy) by recently deceased individuals who gave their brains. What did they uncover, then? In persons with depression, the brain’s microglial cells, a specific type of immune cell, are less active. Contrary to expectations, immune cells are repressed, which is the reverse of inflammation.
Microglial cells maintain synapses, helping neurons communicate efficiently. They also continually scan the central nervous system for damaged neurons, synapses, and pathogens.
Only the microglial cells close to the neurons in the samples from depressed individuals exhibited decreased activity. Therefore, the group examined whether microglial cells receive signals from neurons that cause them to become less active when a person is depressed. And this did come to pass.
Scientists used fresh tissue immediately after death to isolate microglia during the study. They then compared the tissues between depressed people and controls.
Karel Scheepstra (researcher involved in the study and working as a psychiatrist at Amsterdam UMC): “We saw abnormal microglia in depressed patients, with the greatest abnormalities seen in patients who were most depressed just before death. Interestingly, abnormalities were only seen in the gray matter and not in the brain’s white matter. This suggests a likely interaction between the microglia and the structures located in the gray matter: the neurons and synapses.”
“We also looked at the type of alterations. We’ve hypothesized for years that depression is associated with brain inflammation, but we’re now seeing just the opposite: not neuroinflammation, but rather an immune-suppressed type of microglia.”
“We termed them ‘depressed microglia’ and wondered how this is possible. The proteins CD200 and CD47 are located in brain cells and synapses. They interact with microglia and are, as it were, a kind of ‘don’t eat me signal.’ We saw that these proteins were elevated, resulting in suppressed microglia, possibly preventing them from clearing damaged connections.”
“Depression is thought to have something to do with a change in neuroplasticity: the ability to make new connections between neurons. A relatively new antidepressant is ketamine, which intervenes in this process and ensures that more connections grow again. In this study, we show that there is a disturbed neuron-microglia interaction. The next step would be to see what exactly the consequences of the inactive microglia are for the maintenance and formation of connections between neurons.”
“Knowing where things go wrong in the process can provide targets for new medication. Can we make these microglia more active again? And what effect does this have on the course of the disease? For now, we have shown that the brains of people who were depressed during life show altered cell activity. This gives us a better understanding of what goes wrong, which we can then build on.”
Journal Reference:
- Karel W.F. Scheepstra, Mark R. Mizee et al. Microglia transcriptional profiling in major depressive disorder shows inhibition of cortical grey matter microglia. Biological Psychiatry. DOI: 10.1016/j.biopsych.2023.04.020