Microglia are a type of neuroglia (glial cell) located throughout the brain and spinal cord. It accounts for 10–15% of all cells found within the brain.
The cells in microglia protect the brain from injury and infection and are critical during brain development. They help disease—showing up, for example, around brain plaques in people with Alzheimer’s.
A new study has suggested that microglia aren’t monolithic. They come in different flavors, and unlike the brain’s neurons, they’re always changing.

Tim Hammond, Ph.D., a neuroscientist in the Stevens lab at Boston Children’s Hospital said, “This is an ambitious study, perhaps the most comprehensive survey of microglia ever conducted. Up until now, we didn’t have a good way of classifying microglia. We could only say how branched they look, how dense they look under a microscope. We wanted to get an idea of what microglia were doing and ‘thinking.”
Scientists conducted the study on mice. They sequenced RNA using technique Drop-seq from more than 76,000 individual microglia to see which genes were turned on or off. They then sampled from all over the brain and throughout the animals’ lifespan (starting before birth), as well after acute brain injury.
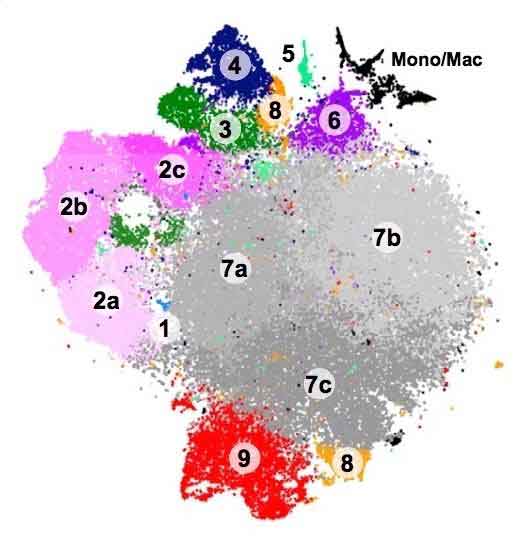
The genetic ‘signatures’ allowed scientists to classify the microglia into at least nine distinct groups, including some types never detected in the past. Some types appeared almost exclusively in the embryonic or newborn stages, others only after injury.
Tim Hammond, Ph.D., a neuroscientist in the Stevens lab at Boston Children’s Hospital said, “The signatures also tell as something about what these cells are doing. If we see microglia in disease, for example, we can begin to parse out: Are they contributing to the disease or are they trying to repair the brain? We think this will help uncover new and interesting roles for microglia that weren’t known before.”
Scientists then classified on a map of the brain, to see how the different varieties of microglia were distributed spatially.
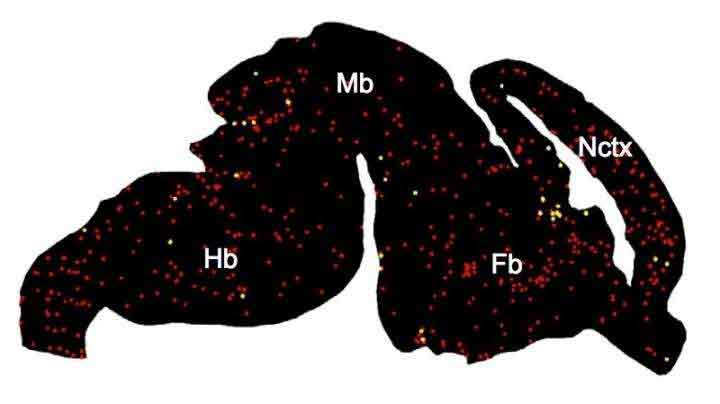
This yielded some fascinating examples. One gathering microglia, for instance (group 4 in the schematic above), would in general group close to the brain’s developing white matter. This recommends they could be associated with myelination, in which nerve fibers are given a layer of protection to enable them to convey motions over longer distances.
Hammond said, “We don’t see those microglia at any other time point or area of the brain. We think they could be important to how the white matter develops, and how axons connect to different parts of the brain.”
“Another tiny but important microglial population (group 8 in the schematic) came to light in the disease setting. The team found it first in a mouse model mimicking multiple sclerosis, which involves a loss of myelination, and later in brain tissue from actual patients with MS.”
“These microglia are very inflammatory compared with normal microglia. It could be a pathological subset that we normally wouldn’t see, but because we sequenced so many microglia we were able to detect this small population.”
“Overall, microglia were most diverse early in brain development, in the aged brain and in disease. The researchers think these distinct groups may shed light on what the cells are doing, and what local cues they’re responding to.”
All this information should help scientists sort out the “good” from the “bad” when it comes to microglia, particularly in so-called activated microglia that appear after brain injury and in diseases like autism and Alzheimer’s. This could help direct the development of drugs to promote the beneficial microglia subsets and block the detrimental ones.
The study is detailed in the journal Immunity.