Biological age, distinct from an individual’s chronological age, has been studied extensively through predictive aging clocks. However, these clocks have limited accuracy in short time scales.
To determine an individual’s biological age, which is different from their chronological age, researchers have created “aging clocks” based on markers such as blood proteins or DNA methylation. The risk of age-related disorders can be forecasted using these aging clocks. Yet, because protein and methylation indicators call for a blood test, less intrusive methods to find similar measures may make aging data more widely available.
Unexpectedly, our retinal features reveal a lot about who we are. Pictures of the retina, connected to the brain via blood vessels, are an essential source of biological and physiological data. Its characteristics have been connected to several age-related disorders, including Alzheimer’s disease, cardiovascular disease, and diabetic retinopathy.
In this new study, scientists showed that deep learning models could accurately predict biological age from a retinal image and reveal insights that better predict age-related disease in individuals. The model is expected to improve the understanding of how genetic factors influence aging.
In its official blog, Google announced that they are releasing code modifications for these models, which build on ML frameworks for analyzing retina images.
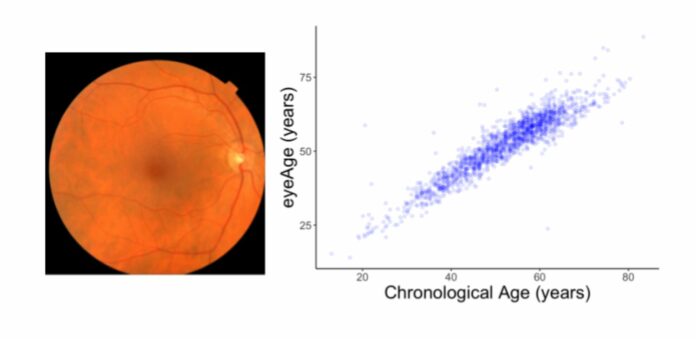
For this study, scientists trained models to predict chronological age using fundus images from the EyePACS dataset to predict individuals’ chronological age. This new ‘aging clocking,’ called ‘eyeAge,’ predicted chronological age more accurately than other aging clocks.
Scientists then evaluated the resulting model performance on a held-out set of 50,000 retinal images and a separate UKBiobank dataset containing approximately 120,000 images. The projections of the eyeAge model closely match people’s actual chronological ages (shown below; Pearson correlation coefficient of 0.87). This is the first time a precise aging clock has been produced using retinal pictures.
Despite the fact that eyeAge and chronological age often correspond well across samples, the figure also depicts individuals whose eyeAge and chronological age are significantly different, depending on whether the model predicts a value that is considerably younger or older. This might mean that the model is taking into account aspects of retinal images that represent actual biological consequences pertinent to the diseases that are increasingly common as people age biologically.
Researchers looked at the association between this discrepancy and diseases like chronic obstructive pulmonary disease (COPD), myocardial infarction, and other health biomarkers like systolic blood pressure to see if underlying biological variables caused it. They found that in these situations, a predicted age that is older than the actual age correlates with illness and health biomarkers. For instance, they demonstrated a statistically significant association between eyeAge and all-cause mortality (p=0.0028), indicating that a higher eyeAge was linked to a higher risk of passing away during the study.
The individual-specific nature of eyeAge was reinforced via multiple GWAS hits in the UK Biobank cohort. The top GWAS locus was further validated via knockdown of the fly homolog, Alk, which slowed the age-related decline in vision in flies.
Researchers noted, “This study demonstrates the potential utility of a retinal aging clock for studying aging and age-related diseases and quantitatively measuring aging on concise time scales, opening avenues for quick and actionable evaluation of gero-protective therapeutics.”
“Our eyeAge clock has many potential applications. As demonstrated above, it enables researchers to discover markers for aging and age-related diseases and to identify genes whose functions might be changed by drugs to promote healthier aging. It may also help researchers further understand the effects of lifestyle habits and interventions such as exercise, diet, and medication on an individual’s biological aging.”
“Additionally, the eyeAge clock could be useful in the pharmaceutical industry for evaluating rejuvenation and anti-aging therapies. By tracking changes in the retina over time, researchers may be able to determine the effectiveness of these interventions in slowing or reversing the aging process.”
“Our findings also indicate that the eyeAge clock, based on retinal images, is independent of blood-biomarker–based aging clocks. This allows researchers to study aging through another angle and, when combined with other markers, provides a more comprehensive understanding of an individual’s biological age. Also, unlike current aging clocks, the less invasive nature of imaging (compared to blood tests) might enable eyeAge to be used for actionable biological and behavioral interventions.”
Journal Reference:
- Sara Ahadi, Kenneth A Wilson et al. Longitudinal fundus imaging and its genome-wide association analysis provide evidence for a human retinal aging clock. eLife. DOI: 10.7554/eLife.82364