The entanglement of two microscopic systems causes their properties to be linked. Manipulating this uniquely quantum phenomenon allows for cryptography, communication, and computation.
Although comparisons between quantum entanglement and the classical physics of heat have been made, the current study shows the limitations of these comparisons. Entanglement is significantly more complex than we previously thought.
The second law of thermodynamics is one of the few physical rules that can be said to be absolutely and undeniably true. According to the law, any closed system’s ‘entropy,’ a physical characteristic, can never diminish. Everyday events are given an “arrow of time” to indicate which processes are reversible and which are not. It explains why an ice cube placed on a hot stove will always melt and why when a valve is opened to the atmosphere, compressed gas will always fly out of its container (and never come back in).
Only states with equal entropy and energy can change from one to the other in a reversible manner. The discovery of thermodynamic processes like the (idealized) Carnot cycle, which places a cap on how effectively one can convert heat into work or the other way around by cycling a closed system through various temperatures and pressures, was made possible by the reversibility condition. Our comprehension of this process was the foundation for the Western Industrial Revolution’s tremendous economic growth.
The second law of thermodynamics applies to any macroscopic system, regardless of the microscopic details. Entanglement, a quantum connection that causes isolated components of the system to share features, may be one of these details in quantum systems. Interestingly, despite the fact that quantum systems are often investigated in the microscopic realm, thermodynamics, and quantum entanglement exhibit many striking parallels. Scientists have discovered a concept known as “entanglement entropy” that, at least for idealized quantum systems completely isolated from their environment, precisely matches the function of thermodynamical entropy.
Quantum information researcher Ludovico Lami said, “Quantum entanglement is a key resource that underlies much of the power of future quantum computers. To make effective use of it, we need to learn how to manipulate it. A fundamental question became whether entanglement can always be reversibly manipulated, in direct analogy to the Carnot cycle. Crucially, this reversibility would need to hold, at least in theory, even for noisy (‘mixed’) quantum systems that have not been kept perfectly isolated from their environment.”
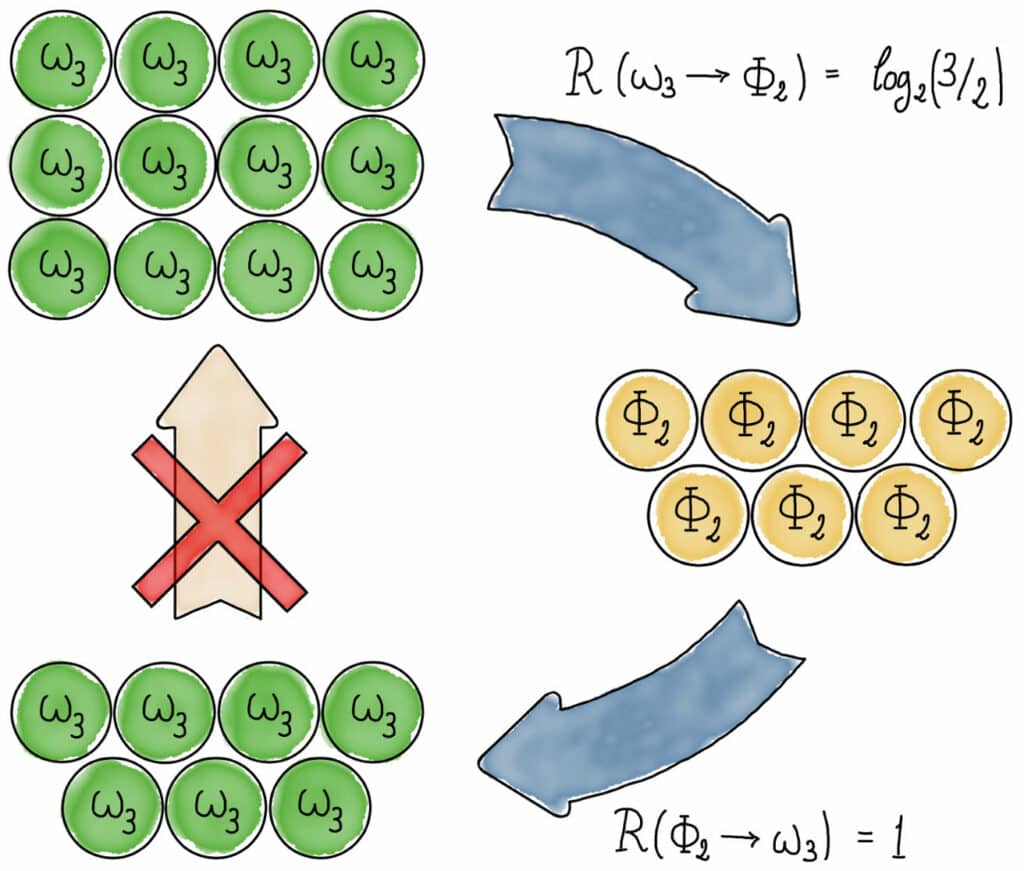
Resolving this long-standing open question, scientists demonstrate that manipulating entanglement is fundamentally irreversible, putting to rest any hopes of establishing a second law of entanglement.
The development of a certain quantum state, which is exceedingly “expensive” to establish with pure entanglement, is necessary for this new conclusion. As the invested entanglement cannot be recovered, there will always be some loss of this entanglement when this condition is created. As a result, changing this condition from one to another and vice versa is essentially impossible. Such states had not before been recognized to exist.
Lami explains: “Because the approach used here does not presuppose what exact transformation protocols are used, it rules out the reversibility of entanglement in all possible settings. It applies to all protocols, assuming they don’t generate new entanglement. Using entangling operations would be like running a distillery where alcohol from elsewhere is secretly added to the beverage.”
“We can conclude that no single quantity, such as the entanglement entropy, can tell us everything there is to know about the allowed transformations of entangled physical systems. The theory of entanglement and thermodynamics are thus governed by fundamentally different and incompatible laws.”
“This may mean that describing quantum entanglement is not as simple as scientists had hoped. Rather than being a drawback, however, the vastly greater complexity of the theory of entanglement compared to the classical laws of thermodynamics may allow us to use entanglement to achieve feats that would otherwise be completely inconceivable. “For now, what we know for certain is that entanglement hides an even richer and more complicated structure that we had given it credit for.”
Journal Reference:
- Lami, L., Regula, B. No second law of entanglement manipulation after all. Nat. Phys. 19, 184–189 (2023). DOI: 10.1038/s41567-022-01873-9