Neurons require controlled polarisation and synaptic material travel to preserve their individual structure and electrical characteristics. Indeed, axonal transport disruption has been linked to various neurodevelopmental and neurological disorders.
According to a new study, the structural integrity of a neuron’s long branch, known as an axon, may require the help of a surrounding protein known as Perlecan.
Researchers discovered that in Drosophila, fruit flies lack perlecan protein. Axonal segments can break apart during development, resulting in the death of synapses.
The extracellular matrix, which is made up of the proteins and other molecules that surround cells, is made more stable and flexible by Perlecan, enabling cells to grow and function in a setting that is encouraging but not constraining.
Study senior author Troy Littleton, the Menicon Professor in MIT‘s Biology and Brain and Cognitive Sciences departments, said, “What we found was that the extracellular matrix around nerves was being altered and essentially causing the nerves to break completely. Broken nerves eventually led to the synapses retracting.”
Humans require at least some Perlecan to survive after birth. Mutations that diminish but do not eliminate Perlecan can result in Schwartz-Jampel syndrome, which causes neuromuscular and skeletal defects.
According to Littleton, the new work could assist in explaining how neurons are harmed in the illness and enhance scientists’ understanding of how the extracellular matrix supports axon and neural circuit development.
The authors discovered that Perlecan was pronounced in a component called the neural lamella, which surrounds axon bundles and works like a rubbery coating around a TV wire to keep the structure intact.
The researchers used serial intravital imaging, a daily imaging of fly brain development, to observe what happened to fly axons and synapses over four days.
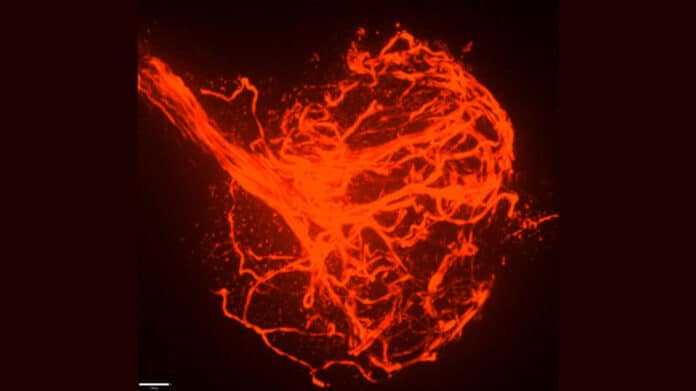
They discovered that while flying axons and synapses initially formed correctly, not only synapses but also entire segments of axons went away. The further an axon segment reached from the fly’s brain, the more likely it was to break apart, implying that the axon segments got more fragile as they stretched further out.
Littleton said, “The breakages were happening in a segment-wide manner. In some segments, the nerves would break, and in some, they wouldn’t. Whenever there was a breakage event, you would see all the neuromuscular junctions (synapses) across all the muscles in that segment retract.”
When scientists compared the structure of the lamella in mutant and healthy flies, they discovered that the mutant lamella was thinner and faulty. Furthermore, where the lamella was weakened, axons were prone to breaking, and the microtubule structures that travel the length of the axon became misdirected, projecting outward and tangled up in spectacular bundles at sites of cut axons.
The researchers demonstrated that Perlecan’s vital significance depended on its secretion from several cells, not just neurons. Blocking the protein in just one cell type or another did not create the same issues as total knockdown, and increasing secretion from neurons alone was insufficient to compensate for its shortage from other sources.
In conclusion, the evidence indicates that a lack of perlecan secretion caused the neural lamella to be thin and faulty, resulting in an overly stiff extracellular matrix.
As the animal moves and pulls on those nerves over time, the loss of flexibility becomes critical. The extracellular matrix is effective early on and can promote development. However, it lacks the characteristics to sustain some critical tasks when the animal begins to move and navigate.
The National Institutes of Health provided funding for the study.
Journal Reference:
- Ellen J, GussYulia Akbergenova et al. Loss of the extracellular matrix protein Perlecan disrupts axonal and synaptic stability during Drosophila development. Neuroscience.DOI: 10.7554/eLife.88273.1