Breast cancer is the most common cancer in women worldwide and the second most common cancer overall. In 2019, approximately 268,600 new cases of invasive and 62,930 new cases of non-invasive breast cancer are expected to be diagnosed in U.S. women. And the sad part is about an estimated 41,760 women will die from breast cancer, according to the American Cancer Society report.
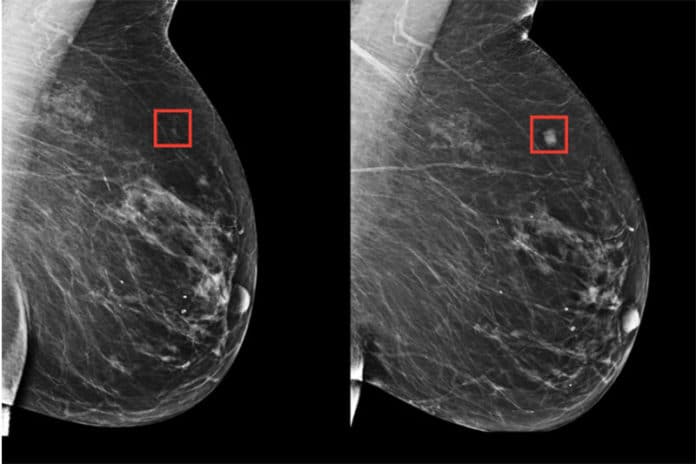
The most common process for diagnosing and screening breast cancer is the mammogram test. In this process, the test is made using the radiations which are the major concern. So the doctors more often advised mammogram after the age of 50. Also, they are not always reliable. About 10% to 15% of women who undergo a mammogram are asked to return following an inconclusive analysis.
Fortunately, addressing all these problems, scientists at MIT’s Computer Science and Artificial Intelligence Laboratory (CSAIL) and Massachusetts General Hospital (MGH) invented a new AI-driven way which helps to detect breast cancer in women for up to five years in advance. This could be the best step towards more consistent and reliable screening procedures.
The team created a new deep learning model that can predict from a mammogram if a patient is likely to develop breast cancer in the future. And the best part is, unlike the older method, it works equally accurate for white and black women.
“Rather than taking a one-size fit all approach, we can personalize screening around a woman’s risk of developing cancer,” says Barzilay, senior author of a new paper in Radiology about the project. “For example, a doctor might recommend supplemental MRI screening for women with high model-assessed risk.”
Instead of manually identifying the patterns, the researchers trained their model on 90,000 mammograms and known outcomes from over 60,000 patients treated at MGH. The model learned the subtle patterns in breast tissue that are precursors to malignancy. This new model was remarkably better than the existing one at predicting the risk of cancer.
“Since the 1960s radiologists have noticed that women have unique and widely variable patterns of breast tissue visible on the mammogram,” said Lehman. “These patterns can represent the influence of genetics, hormones, pregnancy, lactation, diet, weight loss and weight gain. We can now leverage this detailed information to be more precise in our risk assessment at the individual level.”
This is a major step towards making a model for all the women, especially for African American women, who are 43% more likely to die from breast cancer than white women.
“It’s particularly striking that the model performs equally as well for black and white people, which has not been the case with prior risk assessment tools”, says Allison Kurian, associate professor of Medicine and Health Research and Policy at Stanford University School of Medicine. “If validated and made available for widespread use, this could really improve on our current strategies to estimate risk.”
Most breast-cancer risk models focus on a range of risk factors such as age, family history of breast and ovarian cancer, hormonal and reproductive factors, and breast density. But some of these factors are less correlated with breast cancer than others, harming the models’ accuracy.
“The recent federal legislation that requires all women to be informed of their mammographic breast density is a strong message from Congress that women deserve access to their health information,” says Lehman. “We are eager to provide this power of information to all women undergoing screening mammography, not only by sharing their mammographic density but also their future risk of breast cancer.”
The researchers model could be very useful to see if patients are at a greater risk for other health problems, like cardiovascular disease or other cancers.
“Our goal is to make these advancements a part of the standard of care,” says Yala. “By predicting who will develop cancer in the future, we can catch cancer before symptoms ever arise and hopefully save lives.”