The extracellular matrix’s composition, flexibility, and organization inside the central nervous system influence the structure and operation of the brain. Soft biomaterials are required from the perspective of in vitro modeling to replicate the 3D neuronal microenvironments. Although 3D culture and neural network creation in bulk hydrogel systems have been extensively studied, these techniques have limited ability to place cells to simulate complex brain architectures.
Researchers from Monash University Engineering have successfully printed 3D neural networks that can transmit and react to nerve signals using “bioinks” made of living nerve cells (neurons).
Scientists used a tissue engineering approach and bioprinting with two bioinks containing living cells and non-cell materials, respectively. Through this, they could mimic the arrangement of grey matter and white matter seen in the brain.
Professor John Forsythe of the Department of Materials Science and Engineering, who is leading the research, said, “while two-dimensional nerve cell cultures have previously been used to study the formation of nerve networks and disease mechanisms, those relatively flat structures don’t reflect the way neurons grow and interact with their surroundings.”
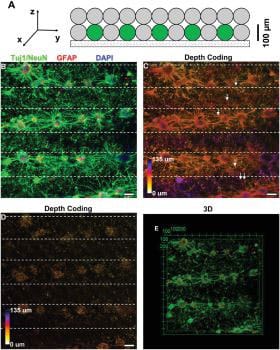
“The networks grown in this research closely replicated the 3D nature of circuits in a living brain, where nerve cells extend processes called neurites to form connections between different layers of the cortex.”
“We found that the projections growing from neurons in the printed ‘grey matter’ or cellular layer readily grew through the ‘white matter’ layer and used it as a ‘highway’ to communicate with neurons in other layers.”
“Not only were we able to construct a basic layout similar to what we see in regions of the brain, we found that the neurons behaved and performed similarly.”
Sensitive electrophysiological tests indicated that in addition to the responses elicited by electrical and pharmacological stimulation, spontaneous nerve-like activity was also occurring in the 3D neural networks.
In neurology and bioprinting, the discovery of observable electrical activity in tissue-engineered 3D networks constitutes a significant advancement.
The formation and growth of nerves and nerve networks, the effects of various disorders on neurotransmission, and the effects of various medications on nerve cells and the nervous system may all be studied using bioprinted 3D neural networks.
Journal Reference:
- Yue Yao, Harold A. Coleman, Laurence Meagher, John S. Forsythe, Helena Parkington. 3D Functional Neuronal Networks in Free-Standing Bioprinted Hydrogel Constructs. Advanced Healthcare Materials. DOI: 10.1002/adhm.202300801