Cells need to compartmentalize thousands of distinct proteins, but the nanoscale spatial relationship of many proteins to the overall intracellular ultrastructure remains poorly understood.
For quite a long time, scientists have explored the tiny world inside cells, developing new instruments to see these basic units of life. In any case, each tool accompanies a tradeoff. Light microscopy, for example, makes it easy to identify explicit cell structures by tagging them with pure to-see fluorescent particles. With the development of super-resolution (SR) fluorescence microscopy, these structures can be seen with even more greater clarity.
In any case, fluorescence can uncover only a few of the more than 10,000 proteins in a cell at a given time, making it difficult to see how these few relate to everything else. Electron microscopy (EM), then again, uncovers every cell structure in high-resolution pictures – however, depicting one element from all others by EM alone can be troublesome as the space inside cells is so crowded.
Scientists thus decided to take the benefit of both of these tools by combining them and creating a new microscopy technique called cryo-SR/EM. Using the method, scientists have captured exquisitely detailed views of the intricate innards of cells, all in 3D.
Using cryo-SR/EM, scientists were able to see tentacled vesicles shuttle cargo for sorting inside the cells. There were also neighboring neurons clinging to one another through a web-like interface, and DNA rearranges in the nucleus as stem cells differentiate into neurons.
Harald Hess, a senior group leader at the Howard Hughes Medical Institute’s Janelia Research Campus, said, “Combining the two techniques gives scientists a clear picture of how specific cellular features relate to their surroundings. This is a very powerful method.”
Primarily, scientists froze the cells under high pressure. Freezing the cells causes cell activity to halt quickly and thus prevents the formation of ice crystals that can damage cells and disrupt the structures being imaged.
Next, scientists place the samples in a cryogenic chamber, where they’re imaged in 3-D by super-resolution fluorescence microscopy at temperatures less than ten degrees above absolute zero. Then, they’re removed, embedded in resin, and imaged in a powerful electron microscope.
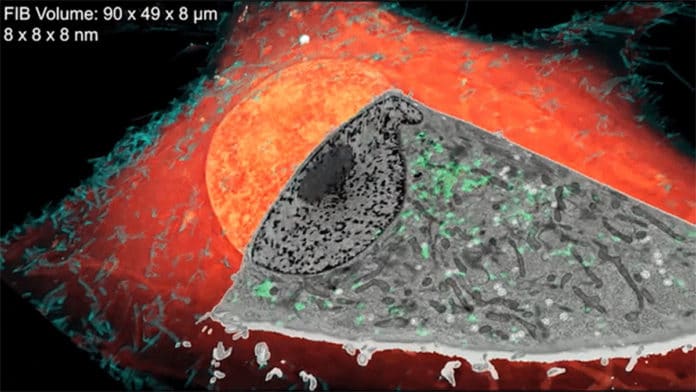
This microscope shoots a beam of ions at the cells’ surface, processing a little bit ceaselessly at a time while taking photos of each recently uncovered layer. A computer program then stitches the pictures together into a 3-D reconstruction.
Finally, researchers overlay the 3-D image data from both microscopes — the result: stunning imagery that reveals cells’ inner details with remarkable clarity.
The study is published in the journal Science.