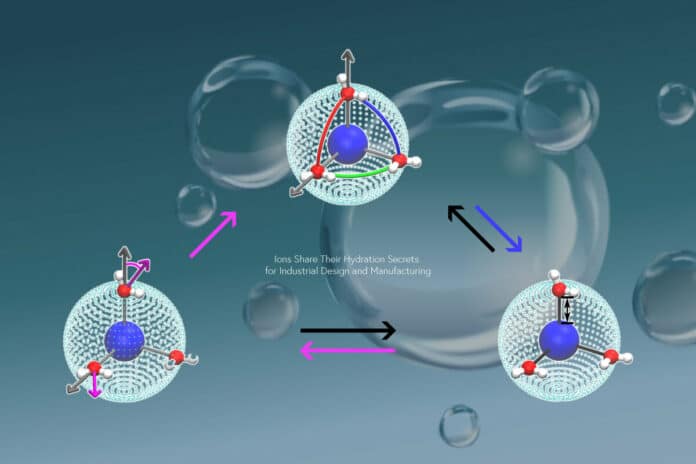
Ions exhibit highly ion-specific complex behaviors when solvated in water, which remains a mystery despite the fundamental importance of ion solvation in nature, science, and technology. A new study explains these ion-specific properties by the ion-induced hierarchical dipolar, translational, and bond-orientational orderings of ion hydration shell under the competition between ion-water electrostatic interactions and inter-water hydrogen bonding.
Scientists from the Institute of Industrial Science, The University of Tokyo, have proposed an explanation for the ion-specific properties of ion hydration in water-based solutions. They have expanded a wide range of applications spanning numerous disciplines, such as chemistry, biology, and materials science, by exposing the nature of ion hydration and how it may be drastically stabilized.
Researchers have long divided salt ions into structure-forming (enabling the creation of water molecule networks) and structure-breaking (destroying these networks) to understand salt ion hydration better. However, a new molecular-level explanation of ion hydration is required in light of recent evidence that this theory needs to be simplified.
Prior research focused on the ionic field, which depends on the ionic charge and the ion-water distance relationship, as a potential driving factor for ion hydration to derive such an explanation. Instead of manufactured or artificial ions, these studies have concentrated on ions with definite sizes and charges found in nature.
Rui Shi, lead author, said, “Our molecular dynamics simulations focus on pseudo-main-group cations that interact with water through two intermolecular forces, Coulomb and Van der Waals interactions.”
“Our work reveals previously unknown details of the ion hydration shell and their impact on water dynamics.”
The study’s key finding is that water molecules interact with ions with lower charge densities more frequently because these molecules form weak bonds with one another. This outcome is independent of the ion. The transition between accelerating and decelerating water dynamics surrounding the ion can be seen as the distance at which the ion-water contact is as strong as water-water hydrogen bonding.
Hajime Tanaka, senior author, said, “We also reveal an explanation for the 11 orders of magnitude difference in the residence times of hydration water. By tuning the ion size and charge to specific combinations, we realize the bond-orientational order of hydrated water molecules can develop and stabilize the hydration shell. This leads us to establish a new mechanism for the highly stable hydration of certain ions.”
This research has numerous applications in chemistry, biology, materials science, and several other sectors. For instance, modern understandings of the processes underlying ion hydration in water-based media may be beneficial for research on exploiting salt hydrates for energy storage and purification technologies for RNA-based medical therapies.
Journal Reference:
- Shi, R., Cooper, A.J. & Tanaka, H. Impact of hierarchical water dipole orderings on the dynamics of aqueous salt solutions. Nat Commun 14, 4616 (2023). DOI: 10.1038/s41467-023-40278-x