Biogas produced by microorganisms present in food waste- consists mainly of methane that can be used for low-cost energy. When methane is converted into ethylene, it can acquire a large added value.
Ethylene is a basic raw material for industries through chemical reactions. It is a typical non-petroleum product that can reduce greenhouse gases.
In 2021, scientists developed a process technology that produces ethylene from Biogas with the help of catalysts.
Biogas contains a significant amount of toxic hydrogen sulfide, which is difficult to remove. It also interferes with the catalytic reaction in ethylene production.
Scientists at the Korea Institute of Science and Technology developed a technology that allows ethylene production by oxidizing and removing hydrogen sulfide during the production process. They then developed a catalyst to improve the reaction activity of ethylene production from Biogas and methane.
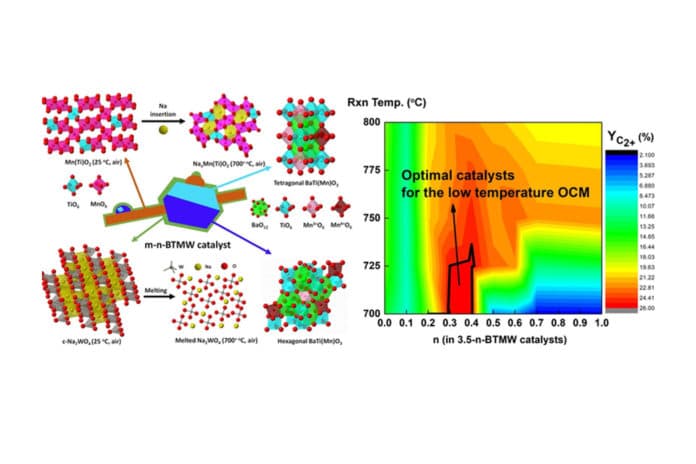
The catalyst is highly resistant to hydrogen sulfide, therefore, does not require hydrogen sulfide removal from Biogas while minimizing the operating temperature from 800oC to 700oC. Through such a reaction, it is possible to produce ethylene from Biogas that contains hydrogen sulfide directly.
Dr. Jeong-Myeong Ha stated, “Biogas is already produced in large quantities in Korea, and if Biogas is used as a raw material for the chemical industry rather than just for heating, biogas producers who struggle to achieve carbon neutrality will have a larger market and be able to provide new raw materials without greenhouse gas emissions. This technology will draw attention from related companies as it can utilize not only Biogas but also methane generated from various wastes such as plastics.”
Journal Reference:
- Lien Thi Do et al. Hybrid catalysts containing Ba, Ti, Mn, Na, and W for the low-temperature oxidative coupling of methane. DOI: 10.1016/j.apcatb.2021.120553