Photocatalysis is a promising technology for generating fuels from sunlight. The obstacle to producing these solar fuels cheaply and abundantly, though, is that they require semiconductors that are efficient but prone to corrosion.
In a recent study, scientists from the Yale School of Engineering & Applied Science have made a breakthrough- that overcomes this challenge. They have found a solution with a first-of-its-kind coating.
Water-splitting systems that use semiconductor materials effectively convert solar energy to chemical energy. Despite capturing sunlight more efficiently, these materials corrode under illumination via self-reduction or self-oxidation.
Protecting these materials adds a limitation to their abilities to separate the charges of negative electrons and positive holes, a process that is essential to photocatalysis but harder to achieve than it is for other systems, such as solar cells. Ordinarily, layers intended to guard these systems protect just one of the systems’ two electrodes, particularly the cathode or anode, restricting it to allow transport of one or the other electron or hole– yet not both.
Shu Hu, assistant professor of Chemical & Environmental Engineering, said, “But to make this new type of photocatalysts for the coevolution of two reactions, we knew that we needed to make the two charges generated within the light-absorbing material (known as the photoabsorber) transport through the coating to reach the reductive and oxidative reaction sites – in which electrons are either added or removed – both located on the surface.”
Tianshuo Zhao, the lead author of the study, said creating a coating that allows for both reactions would be a big step. Doing so successfully would “boost the solar-to-hydrogen efficiency to an order of magnitude higher.”
“We’ve developed a very effective protection strategy for these narrow bandgap semiconductors, stabilizing them during photoreactions, which requires more than the traditional protective design.”
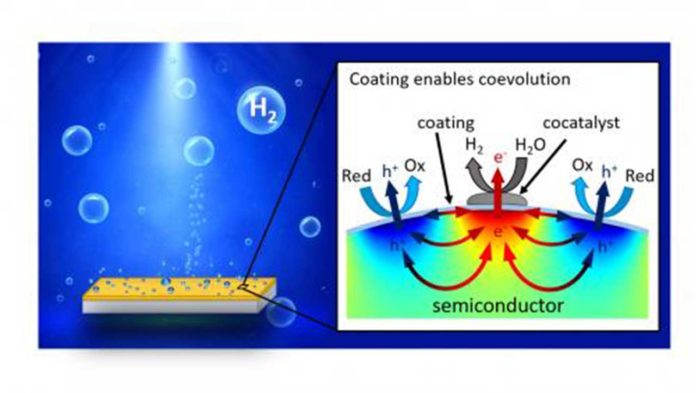
Scientists created a 3-nanometer-thick coating using titanium oxide to encapsulate the photoabsorber constantly. There are metal nanoparticles on the top of the coating that serve as co-catalysts.
The structure allows the photocatalyst to perform different reactions at the cocatalyst site and bare coating surface concurrently. The system is designed to scale up readily. It has achieved a record-high energy-conversion efficiency of 1.7% with narrow-bandgap semiconductors while remaining stable for more than 150 hours, a much longer period than other systems of similar materials.
Hu said, “The combination of coatings and cocatalysts makes it possible for electrons and holes to transport through the coating by design with minimum recombination separately. That makes this kind of particle-based solar fuel generators work.”
Journal Reference:
- Tianshuo Zhao et al. A coating strategy to achieve adequate local charge separation for photocatalytic coevolution. Proceedings of the National Academy of Sciences, 2021; DOI: 10.1073/pnas.2023552118