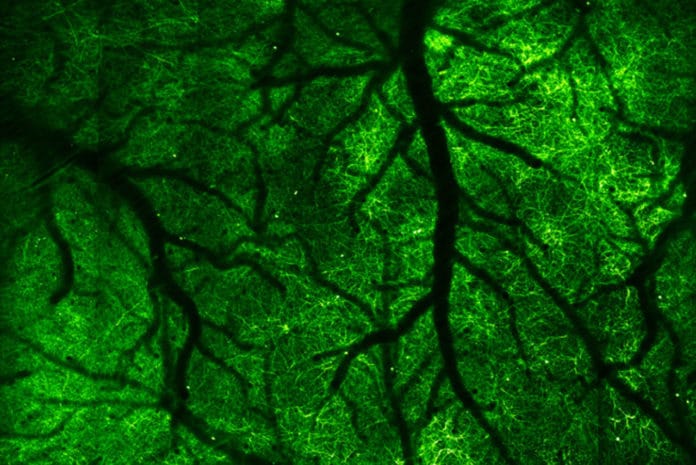
Using a newly developed camera: a microscope, scientists from the University of California, Berkeley have captured electrical and chemical signals flashing through our brains- the neuron fires- as we move through the world.
Scientists imaged the brain of an alert mouse 1,000 times a second. For the first time, they have recorded the passage of millisecond electrical pulses through neurons.
Scientists developed this imaging technique by combining two-photon fluorescence microscopy and all-optical laser scanning in a futuristic microscope that can image a two-dimensional slice through the neocortex of the mouse brain up to 3,000 times per second.
This technique is expected to help neuroscientists to clock electrical signals as they propagate through the brain. They can also figure out the transmission problems associated with the disease.
The additional benefit of this technique is that it allows tracing the hundreds to tens of thousands of inputs any given brain cell receives from other brain cells, including those that don’t trigger the cell to fire. These sub-threshold inputs — either exciting or inhibiting the neuron — gradually add up to a crescendo that triggers the cell to fire an action potential, passing information along to other neurons.
Lead researcher Na Ji, a UC Berkeley associate professor of physics and of molecular and cell biology, said, “The typical method for recording electrical firing in the brain, via electrodes embedded in the tissue, detects only blips from a few neurons as the millisecond voltage changes pass by. The new technique can pinpoint the actual firing neuron and follow the path of the signal, millisecond by millisecond.”
“In diseases, many things are happening, even before you can see neurons firing, like all the subthreshold events. We’ve never looked at how a disease will change with subthreshold input. Now, we have a handle to address that.”
Along with this study, scientists published another study in the same issue, demonstrating a different technique for imaging calcium signaling over much of an entire hemisphere of the mouse brain at once.
Ji said, “This is the first time anyone has shown in three dimensions the neural activity of such a large volume of the brain at once, which is far beyond what electrodes can do. Furthermore, our imaging approach gives us the ability to resolve the synapses of each neuron.”
“In brain disorders, including neurodegenerative disease, it’s not just a single neuron or a few neurons that get sick. So, if you want to understand these illnesses, you want to be able to look at as many neurons as possible over different brain regions. With this method, we can get a much more global picture of what is happening in the brain.”
Scientists can now peer into the brain using probes that can be pinned to specific types of cells and become fluorescent when the environment changes. Scientists even tracked voltage changes in neurons using a sensor developed by co-author Michael Lin of Stanford University that becomes fluorescent when the cell membrane depolarizes as a voltage signal propagates along the cell membrane.
Scientists, later on, enlighten these fluorescent probes with a two-photon laser, which causes them to emit light, or fluoresce, on the off chance that they have been actuated. The emitted light is captured by a microscope and combined into a 2D image that shows the location of the voltage change or the presence of a particular chemical, for example, the signaling ion, calcium.
By rapidly scanning the laser over the brain, much like a flashlight that gradually reveals the scene inside a darkened room, scientists obtained images of a single, thin layer of the neocortex. They then conducted 1,000 to 3,000 full 2D scans of a single brain layer every second by replacing one of the laser’s two rotating mirrors with an optical mirror — a technique called free-space angular-chirp-enhanced delay (FACED).
The kilohertz imaging uncovered millisecond changes in voltage. It also showed slowly changing concentrations of calcium and glutamate, a neurotransmitter, as deep as 350 microns (one-third of a millimeter) from the brain’s surface.
Two-photon fluorescent microscopy was combined with a different technique, Bessel focus scanning to acquire 3D images of the movement of calcium through neurons.
To keep away from lengthy scans of each micron-thick layer of the neocortex, the excitation laser of the two-photon laser is formed from a point to a small cylinder, similar to a pencil, around 100 microns long. This pencil beam is then scanned at six distinct depths through the brain, and the fluorescent images are joined to make a 3D image.
Ji said, “Using conventional methods, we would have to scan 300 images to cover this volume, but with an elongated beam that collapses the volume onto a single plane, we only need to scan six images, which means that now we can have a fast enough volumetric rate to look at its calcium activity.”
Scientists are now working to achieve high-speed, high sensitivity images deep in the neocortex, which is about 1 millimeter thick.
Ji said, “As a way to understand the brain, my dream is to combine these microscopy techniques to get a submicron spatial resolution so we can see the synapses, millisecond time resolution for the voltage imaging, and see all of this deep in the brain. What is complicated and challenging about the brain is that, if you only do one single optical section, in a way, you don’t get a complete picture because a neural network is very much three-dimensional.”
Co-authors on the voltage imaging paper with Ji, Lin, and Tsia are Jianglai Wu and Shuo Chen of UC Berkeley, Yajie Liang, and Ching-Lung Hsu of the Howard Hughes Medical Institute (HHMI) Janelia Research Campus in Virginia, and Mariya Chavarha, Stephen Evans and Dongqing Shi of Stanford.
Co-authors with Ji on the calcium imaging paper are co-first authors Rongwen Lu and Yajie Liang of Janelia and Guanghan Meng of UC Berkeley; Pengcheng Zhou and Liam Paninski of Columbia University; and Karel Svoboda of Janelia.
References:
- Kilohertz two-photon fluorescence microscopy imaging of neural activity in vivo. DOI: 10.1038/s41592-020-0762-7
- Rapid mesoscale volumetric imaging of neural activity with synaptic resolution. DOI: 10.1038/s41592-020-0760-9