Cancer treatments regularly kill both healthy and infected cells, activating wrecking symptoms. Also, some cancers, which from the outset have all the earmarks of being diminished in size after a progression of medicines, reappear in a much progressive form, and impact proposed to be driven by cancer stem cells. Improving exactness and constraining the risk of disease recurrence is, in this manner, two of the fundamental goals of research when developing up any new treatment.
In 2014, Roberto Coppari, Professor in the Department of Cell Physiology and Metabolism at UNIGE Faculty of Medicine, recognized a protein called FKBP10, which is expressed in human lung malignancy cells; however not in healthy cells. This protein has the capability of being significant for quickly dividing cells, and should subsequently be communicated during undeveloped embryonic and in the early stages of life when the developing organism must develop rapidly.
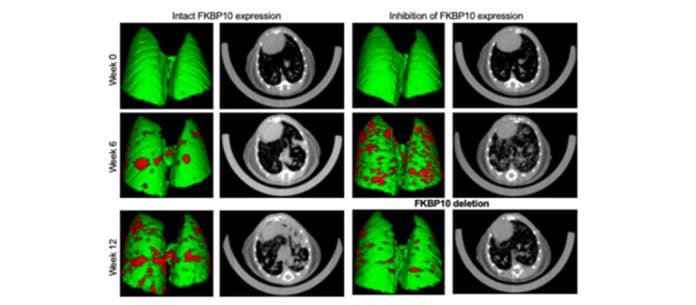
Scientists from the University of Geneva (UNIGE) now tested the role of FKBP10 in cancer progression. For this, they studied a mouse model of lung cancer.
Coppari said, “The inhibition of FKBP10 expression in some of our mice, particularly after tumor development, led to the near disappearance of cancer. This confirms, in a whole organism, the results we had previously obtained with human cancer cells. It also shows, and this is important from a therapeutic point of view, that the process can be reversed after cancer onset.”
Martine Collart, Professor in the Department of Microbiology and Molecular Medicine at UNIGE Faculty of Medicine, whose expertise in protein synthesis and assembly has proved essential said, “Furthermore, FKBP10 also appears to be involved in the high risk of relapse: a few months or years after initial effective treatment, the tumor may return very aggressively. Resistance comes from so-called “cancer stem cells” that have a particular profile.”
“Treatments generally aim at destroying rapidly dividing cells, as are most tumor cells. However, cancer stem cells can escape because they do not divide rapidly. They can be even selected and enriched by chemotherapies before becoming capable of proliferating, and then induce relapse in an even more aggressive form. In this context, FKBP10 appears to be important for protein synthesis and particularly upon the insertion of an amino acid called proline. Hence, by inhibiting FKBP10, we not only arrested the proliferation of cancer cells but also hindered the formation of cancer stem cells.”
Undoubtedly, to have the option to expect their capacity in cells, proteins must overlap accurately after synthesis. This wonder is connected to how the amino acids are organized. One of them, proline, can adopt various setups, and in this way permit proper folding and function of proteins in which it is incorporated, or not. Also, FKBP10 changes the isomerization of proline – how it is arranged – in proteins.
However, not all tumors express FKBP10; this protein does appear in a good number of them, particularly those affecting lungs, colon, or breasts.
In this study, scientists adopted two different but complementary approaches: Roberto Coppari is in the process of reanalyzing dozens of drugs already approved to test their effect on FKBP10, in the hope of identifying an active inhibitory molecule.
Journal Reference:
- FKBP10 Regulates Protein Translation to Sustain Lung Cancer Growth. DOI: 10.1016/j.celrep.2020.02.082