Every single animal cell has an organelle called a centrosome, which is fundamental to the organization of their cell skeleton. The centrosome plays major jobs, particularly during cell division, where it permits the equivalent sharing of genetic data between two daughter cells.
At the point when the cells quit isolating, the centrioles, cylindrical structures made out of microtubules at the base of the centrosome, move to the plasma membrane and permit the development of essential and cell cilia, which are utilized separately for the exchange of data and the genesis of movement.
While performing these pivotal biological functions, centrioles are along these lines exposed to numerous physical powers, which they must resist.
Scientists from the University of Geneva (UNIGE) have discovered an internal structure at the center of these nano-cylinders, a real cellular scaffolding that maintains the physical integrity of this organelle.
They have discovered a new nano-structure that lies at the center of our cellular skeleton. This discovery will allow understanding better how the cell maintains its architecture as well as the pathologies associated with dysfunctions of this structure.
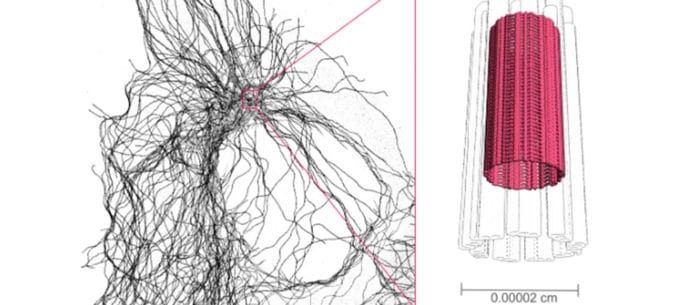
Paul Guichard, Professor in the Department of Cell Biology of the Faculty of Science at UNIGE, said, “Centrioles, formed by microtubules, are components of the cell skeleton. They have a canonical organization defined by nine triplets of microtubules that must be maintained as a structural unit to resist the various forces they face during their cellular functions.”
For this study, scientists used an internal scaffolding for this organelle using high-powered electron microscopes, in collaboration with researchers at the University of Basel and the Helmholtz Campus in Neuherberg, Germany.
Maeva Le Guennec, a UNIGE researcher and first author of the study, said, “This study allowed to analyze centrioles of four different species and to demonstrate that this inner scaffold is present systematically.”
Virginie Hamel, a researcher at the Department of Cell Biology and co-leader of the study, said, “We then investigated which centriolar proteins were located in this new structure. To do so, we used an innovative super-resolution method, called expansion microscopy, which makes it possible to inflate cells without deforming them to observe their internal organization. Thus, we were able to identify four proteins that are located at the level of this inner scaffold.”
Virginie Hamel noted, “We realized that the four proteins we identified are associated with pathologies related to retinal degeneration.”
Paul Guichard said, “The loss of retinal photoreceptors is possibly due to a failure to maintain the microtubule doublets present in these specialized cells. We now intend to discover the possible link between such a structural maintenance defect and retinal disorders, to pave the way for a better understanding of this pathology.”
This study is published in the journal Science Advances.