Cell migration is an essential process that controls development, tissue maintenance, organ health, and functionality. Cells interact with the extracellular matrix (ECM) around them, responding to its chemical and mechanical composition through adhesions, shape, and force generation.
Cancer cells can smell a layer of cells beneath the top collagen layer on which they regularly travel. However, according to new research from Washington University in St. Louis’ McKelvey School of Engineering, normal cells cannot.
According to Amit Pathak, an associate professor of mechanical engineering and materials science, and Christopher Walter, a postdoctoral research associate in Pathak’s lab, Cancer cells have “depth mechanosensing,” or the ability to sense the properties of distant environments beneath their immediate extracellular matrix.
This might mean that the mechanical qualities of the extracellular matrix placed outside of cells can influence cell migration.
Amit Pathak and his colleagues have long studied cell migration to learn how cancerous tumors get into the tissue, wounds heal, and organs and tissues develop.
They previously demonstrated that cells move quicker in stiffer settings due to stronger pressures and tractions than in soft environments.
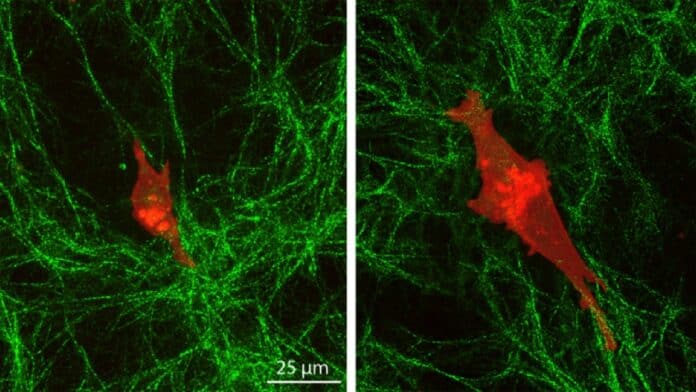
Pathak and Walter developed two hydrogels, one rigid and one soft, and placed a collagen-based extracellular matrix on top. Normal cells changed little in shape or size and migrated slowly on top of the collagen layer.
They observed the behavior of human mammary normal epithelial cells and triple-negative breast cancer cells on the surfaces using a range of techniques, including atomic force microscopy, high-resolution imaging, and cell movement dynamics studies.
Normal cells changed shape and size a little and moved slowly on top of the collagen layer. Because of their deep mechanosensing, cancer cells on the stiff underlying gel elongated and extended more stable protrusions, allowing them to communicate across matrix layers. They also moved faster and created more pulling of collagen fibers.
Pathak said, “Cells sense deep into their environment, and their polarity installs a polarity in the extracellular matrix according to what the cell is telling it. These behaviors suggest that the stiffness of the underlying gel influences the shapes of the cancer cells, while normal cells respond only to the immediate, soft collagen gel. This is something that was not known before.”
Jairaj Mathur, a doctorate student in mechanical engineering and materials science, created a computational model to explain the deep sensing found in studies by integrating polarised forces of cells with collagen remodeling. His model demonstrated that if the cells do not receive feedback from the matrix, a sense of depth may occur briefly but will not be sustained. Cancer cells excel at this because they can recruit the matrix to migrate and sense depth.
According to Walter, the cells’ polarity also helps generate force for the cells to travel in a specific direction. However, they also need the ability to pull on collagen fibers and adapt their surroundings to make them friendlier for migration.
He said, “We were able to get rid of their ability to sense the distant matrix by cutting the collagen itself or cross-linking it, eliminating the feedback that the matrix gives the cells. If you lose either mechanism, the feedback stops, so they have to have both.”
Cancer cells grew and expanded more stable protrusions on the stiff underlying gel, allowing them to communicate across matrix layers and travel more quickly.
Journal Reference:
- Christopher Walter, Jairaj Mathur, et al. Reciprocal intra- and extracellular polarity enable deep mechanosensing through layered matrices. Cell Reports. DOI: 10.1016/j.celrep.2023.112362