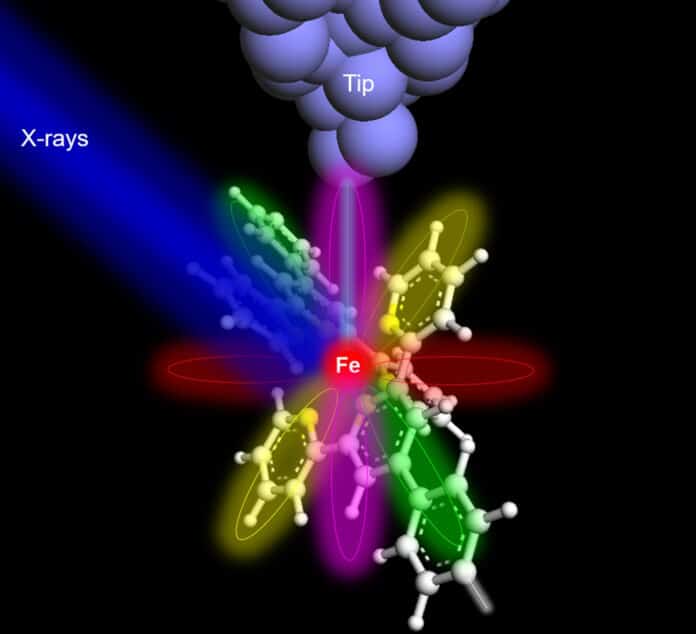
Since the discovery of X-rays, their use has been ubiquitous, from medical and environmental applications to materials sciences. Characterizing X-ray requires several atoms, and reducing the material quantity is a long-standing goal.
A team of scientists from Ohio University, Argonne National Laboratory, the University of Illinois-Chicago, and others, led by Ohio University Professor of Physics and Argonne National Laboratory scientist Saw Wai Hla, have taken the world’s first X-ray SIGNAL (or SIGNATURE) of just one atom.
Scanning probe microscopes can regularly image atoms, but without X-rays, it is impossible to determine what materials they are made of. One bit at a time, deciding on an atom’s specific kind while measuring its chemical state is possible.
Hla said, “Once we can do that, we can trace the materials down to the ultimate limit of just one atom. This will greatly impact environmental and medical sciences and maybe even find a cure that can have a huge impact on humankind. This discovery will transform the world.”
Scientists used a purpose-built synchrotron X-ray instrument at the XTIP beamline of Advanced Photon Source and the Center for Nanoscale Materials at Argonne National Laboratory.
The group used an iron and a terbium atom in their respective molecular hosts as examples. The research team used a technique called synchrotron X-ray scanning tunneling microscopy, or SX-STM, which involves placing a special detector made of a sharp metal tip in close proximity to the sample to collect excited X-ray electrons, in addition to conventional X-ray detectors, to detect the signal of a single atom. The photoabsorption of core-level electrons, which creates elemental fingerprints and effectively identifies the elemental type of the materials, initiates X-ray spectroscopy in SX-STM.
Hla said, “The spectrums are like fingerprints, each unique and able to detect exactly what it is.”
Tolulope Michael Ajayi, the first author of the paper and doing this work as part of his Ph.D. thesis, said, “The technique used, and concept proven in this study, broke new ground in X-ray science and nanoscale studies. More so, using X-rays to detect and characterize individual atoms could revolutionize research and give birth to new technologies in areas such as quantum information and the detection of trace elements in environmental and medical research, to name a few. This achievement also opens the road for advanced materials science instrumentation.”
Hla explained, “We have also detected the chemical states of individual atoms. By comparing the chemical states of an iron atom and a terbium atom inside respective molecular hosts, we find that the terbium atom, a rare-earth metal, is isolated and does not change its chemical state. In contrast, the iron atom strongly interacts with its surrounding.”
With this finding, scientists can now determine not only the type of element but also its chemical state, enabling them to better manage the atoms inside diverse material hosts to satisfy the constantly shifting requirements in various disciplines. Additionally, they have created a brand-new technique known as “X-ray excited resonance tunneling, or X-ERT,” which enables them to use synchrotron X-rays to determine how a single molecule’s orbitals are oriented on a material surface.
Hla said, “This achievement connects synchrotron X-rays with the quantum tunneling process to detect the X-ray signature of an individual atom and opens many exciting research directions, including the research on quantum and spin (magnetic) properties of just one atom using synchrotron X-rays.”
Journal Reference:
- Ajayi, T.M., Shirato, N., Rojas, T. et al. Characterization of just one atom using synchrotron X-rays. Nature 618, 69–73 (2023). DOI: 10.1038/s41586-023-06011-w