A Rutgers University scientists have developed a new “virtual biopsy” device that can quickly determine a skin lesion’s depth and potential malignancy without using a scalpel. The device performs virtual biopsies of skin lesions, by combining optical coherence tomography images with stiffness measurements performed simultaneously using vibrational analysis.
Researchers believe that the ability to analyze a skin tumor non-invasively could make biopsies much less risky and distressing to patients. Currently, physicians who perform surgical biopsies often don’t know the extent of a lesion. Also, they are unaware of whether it will be necessary to refer the patient to a specialist for extensive tissue removal or plastic surgery – until surgery has already begun.
The device called Vibrational Optical Coherence Tomography (VOCT) creates a 3D map of the lesion’s width and depth under the skin using a tiny laser diode. It also uses soundwaves to assess the lesion’s density and stiffness, since tumors and cancer cells are stiffer than healthy tissues.
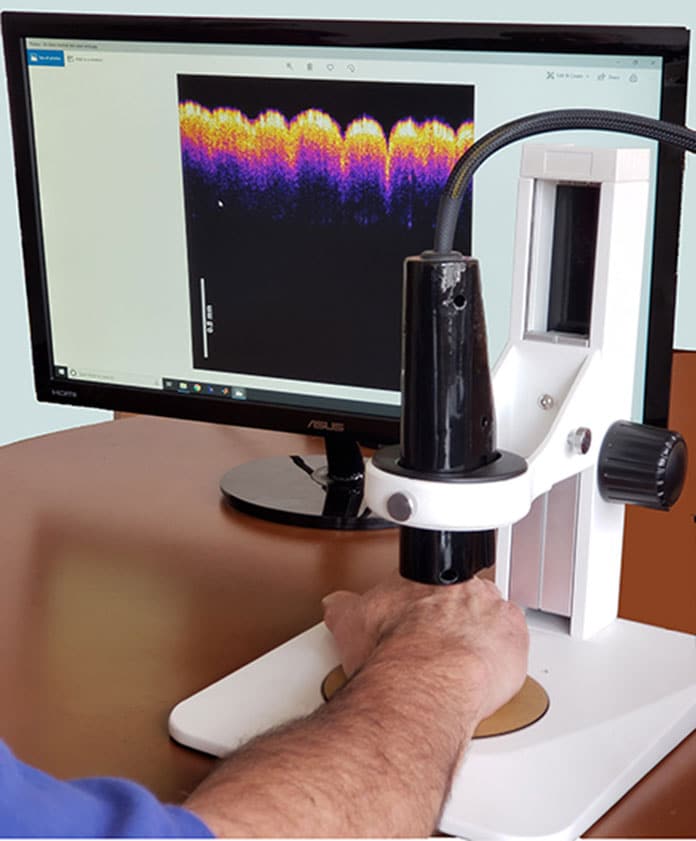
An inch-long speaker applies audible soundwaves against the skin to measure the skin’s vibrations and determine whether the injury is malignant.
“This procedure can be completed in 15 minutes with no discomfort to the patient, who feels no sensation from the light or the nearly inaudible sound,” said lead researcher Frederick Silver, a professor of pathology and laboratory medicine at Rutgers Robert Wood Johnson Medical School. “It’s a significant improvement over surgical biopsies, which are invasive, expensive, and time-consuming.”
The researchers tested the prototype VOCT device over six months on four skin excisions and eight volunteers without skin lesions. They found that precancerous and cancerous lesions were characterized by changes in both the morphology and stiffness of the cellular components of the skin.
The virtual biopsy device could accurately distinguish between healthy skin and different types of skin lesions and carcinomas, according to a report in Wiley Online Library. It is now waiting for FDA approval for large-scale testing.
Further studies are needed to fine-tune the device’s ability to identify a lesion’s borders and areas of highest density and stiffness, which would allow physicians to remove tumors with minimally invasive surgery.