By combining infrared videography with AI, scientists from the University of Rochester have created a new microscopy technique to track microscopic immune cells’ interactions in a living eye without dyes or damage. This new technique is expected to become a game-changer in the field of pharmaceuticals.
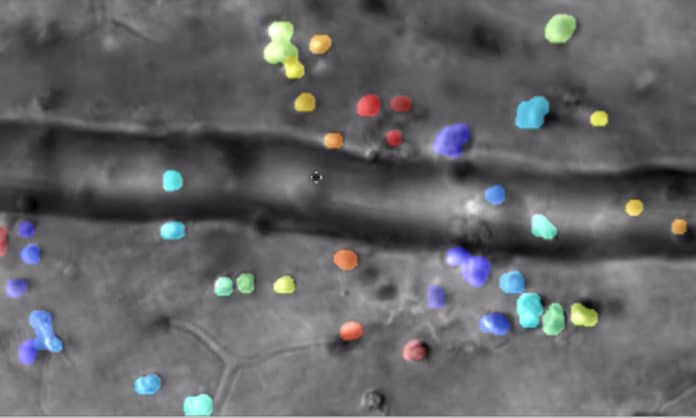
Scientists have captured a video where we can see immune cells moving through living retinal tissue at the back of an eye.
One of the clip shows immune cells crawling slowly along the inside edge of a blood vessel that the video must be sped up 25 times to show their progress. Another cell slowly treads against the flow of blood in a vessel. Other immune cells leave the blood vessels and inch through the surrounding tissue, then congregate in a swarm, forming a beehive of activity.
Using the technique, scientists were able to noninvasively image and track translucent immune cells’ interactions within live retinal tissue in animals.
Rochester vision scientist Jesse Shallek said, “We think of the eye as this beautiful window where we can peer in, noninvasively, without having to cut or insert a camera into places where we would rather it didn’t go. The eye is an extension of our brain, and therefore with this technology, we have some of our first glimpses into immune cells function deep in the central nervous system. This is a critical step forward for basic science and clinical study alike.”
Scientists are now using adapting the new technique for use with human patients.
Schallek said, “We think this will be a game-changer for ophthalmology and for our understanding of retinal diseases that lead to blindness.”
What role do immune cells play in inflammation?
Schallek says, “Immune cells are at the center of “a whole cascade of events” that cause the inflammation that is characteristic of most retinal eye diseases that lead to blindness. For example, in addition to immune cells arriving at the affected tissue, and releasing compounds that recruit more immune cells, there are also changes in blood flow—all of which interfere with vision and complicate the progression of the disease.”
“Until now, the tools available to measure inflammation in retinal tissue have been limited.”
“Optical coherence tomography, for example, has been used to measure the thickness of retinal tissue at the back of the eye. The thickness of that tissue is thought to be a marker for how inflamed the tissue is. However useful that might be, it doesn’t tell you what the cells in that tissue are doing.”
The new technology developed by his lab does that by:
- Building on Rochester‘s adaptive optics technology by David Williams, director of the Center for Visual Science, and his colleagues more than 20 years ago. Adaptive optics provided a way to correct for aberrations of the eye so that researchers could visualize individual cells at the back of the eye for the first time.
- Integrating into adaptive optics, a new phase contrast technique – much like differential interference contrast microscopy–which can capture images of translucent objects such as immune cells.
- Using time-lapsed videography to capture immune cell activity images in the retina over periods ranging from milliseconds to months. Adjusting the playback speed allows the slow movements of those cells can be more easily tracked.
- Using artificial intelligence (AI) computer code deployed by the lab to identify the different kinds of immune cells captured in the images.
Using ultra-high-speed imaging of individual red blood cells to simultaneously track blood flow and how it changes in response to the inflammation.
Colin Chu, an ophthalmologist and visiting senior research fellow from the University of Bristol said, “A defining characteristic of immune cells is that they are truly mobile and incredibly dynamic, rushing to wherever inflammation occurs. The first time we successfully imaged them was astounding, as we were essentially spying on them as they worked within their actual native setting. Even now, watching the recordings continues to mesmerize me.”
Aby Joseph, a Ph.D. candidate at the Institute of Optics, said, “The use of low levels of infrared light to achieve this means that our approach can be safely translated to human study. Additionally, the use of high-speed imaging to measure blood flow revealed surprising details about how inflammation behaves in the central nervous system. We found that veins and arteries increase blood flow to the inflamed retina through markedly different ways. This could become important for designing and testing future treatments to resolve inflammation.”
Other coauthors of the study include Guanping Feng and Kosha Dholakia, also students in Schallek’s lab.
The project was supported with funding from the National Eye Institute of the National Institutes of Health, Research to Prevent Blindness, F. Hoffmann-La Roche Ltd. Roche Academy of Distinguished Scholars, the Dana Foundation, and the World Universities Network WUN Research Mobility Programme award.
Journal Reference:
- Aby Joseph et al. Imaging single-cell blood flow in the smallest to largest vessels in the living retina. DOI: 10.7554/eLife.45077