Lithium metal batteries using solid electrolytes are considered to be the next-generation lithium batteries. These batteries are lightweight, inflammable, pack a lot of energy, and can be recharged very quickly. However, solid electrolytes are susceptible to lithium-metal short-circuiting and failure, and the root cause is under debate.
Now, researchers at Stanford University and SLAC National Accelerator Laboratory say they have finally solved the mystery. The discovery could lead to new designs and battery production methods that help to avoid the problem.
In their rigorous, statistically significant experiments, researchers found that mechanical stress experienced while charging cause solid electrolytes to fail.
“Just modest indentation, bending or twisting of the batteries can cause nanoscopic fissures in the materials to open and lithium to intrude into the solid electrolyte causing it to short circuit,” explained senior author William Chueh. “Even dust or other impurities introduced in manufacturing can generate enough stress to cause failure.”
The problem of failing solid electrolytes has puzzled researchers for years. Some theories say it was due to the unintended flow of electrons, while others point to chemistry.
Now, through more than 60 experiments, the Stanford researchers demonstrated that the problems were, in fact, caused by nanoscopic cracks, dents, and fissures within ceramic electrolytes. Many of these fractures measured less than 20 nanometers wide.
During the fast charging, the team says, these inherent fractures open that allow the lithium to intrude, causing the battery to short circuit or fail.
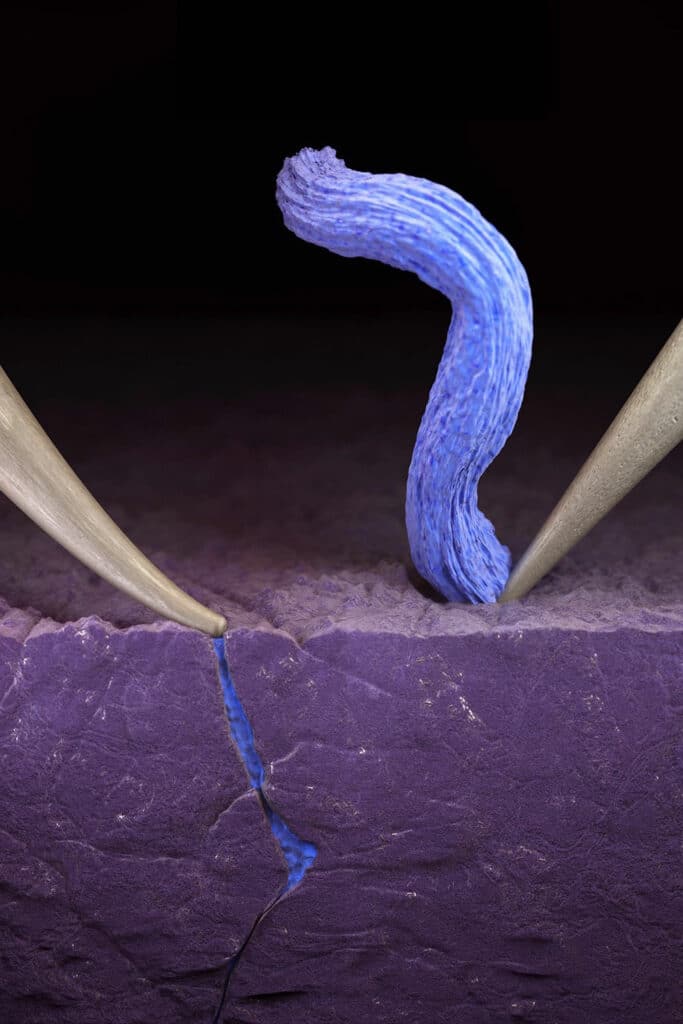
The researchers applied an electrical probe to a solid electrolyte in each experiment, creating a miniature battery. They used an electron microscope to observe fast charging in real-time.
They then used an ion beam as a scalpel to understand why the lithium collects on the surface of the ceramic in some locations, while in other spots, it begins to burrow deeper and deeper – until the lithium bridges across the solid electrolyte, creating a short circuit.
After all the experiments, researchers concluded that the difference is pressure. When the electrical probe merely touches the surface of the electrolyte, lithium gathers beautifully atop the electrolyte even when the battery is charged in less than one minute.
However, when the probe presses into the ceramic electrolyte – mimicking the mechanical stresses of indentation, bending, and twisting – it is more probable that the battery short circuits.
“Given the opportunity to burrow into the electrolyte, the lithium will eventually snake its way through, connecting the cathode and anode,” said McConohy, who completed his doctorate last year working in Chueh’s lab and now works in the industry. “When that happens, the battery fails.”
Using these new findings, the team is now looking at ways to use these very same mechanical forces intentionally to toughen the material during manufacturing. They are also looking at ways to coat the electrolyte surface to prevent cracks or repair them if they emerge.
As a result of this discovery, energy-dense, fast-charging, non-flammable lithium metal batteries that last a long time could overcome the main barriers to the widespread use of electric vehicles, among numerous other benefits.
Journal reference:
- Geoff McConohy, Xin Xu, Teng Cui, Edward Barks, Sunny Wang, Emma Kaeli, Celeste Melamed, X. Wendy Gu, and William C. Chueh. Mechanical regulation of lithium intrusion probability in garnet solid electrolytes. Nature Energy, 2023; DOI: 10.1038/s41560-022-01186-4
