For the first-ever time, scientists at the IBM Research–Zurich, ExxonMobil Research and Engineering Company and Universidade de Santiago de Compostela have successfully imaged molecules as they change their charge states.
It is well known that molecules change their charging state for some time. But, until now, none of the scientists have been able to see them in action.
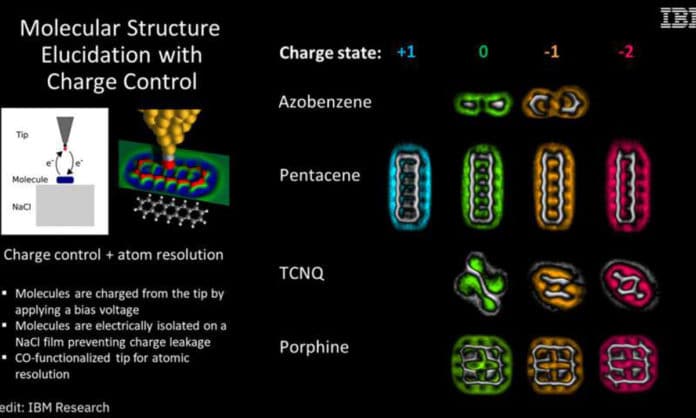
In this new effort, the researchers imaged four molecules—azobenzene, pentacene, TCNQ, and porphine—as they changed due to charging. They note that molecular load is at the very heart of many fundamental biological processes, such as energy transport and photoconversion—thus, observing what it looks like when it happens is very important.
To capture the images, scientists put a single particle on an isolated NaCl film and then used high-resolution nuclear power microscopy in an icy vacuum environment to move a single electron from the probe tip to the atom. Imaging was completed using carbon monoxide-functionalized tips. Each of the fragments was imaged in four states: positive, neutral, negative, and double-negative (including two electrons).
Scientists watched structured changes in the majority of the particles, and that each was changed unexpectedly in comparison to the others. With pentacene, for instance, scientists observed which regions of the molecules turned out to be progressively receptive.
With TCNQ, they observed changes in the bonds between the atoms in the molecule—and they also noted that it moved relative to its base. And with porphine, they found changes to the types of bonds and their lengths.
Scientists also noted that the porphine, in particular, plays a vital role in biological processing—it is involved in the process of hemoglobin transporting oxygen in living organisms.
Being able to see what happens to a molecule as it is charged can help to understand better how the whole transport process works. The team further suggests that using techniques such as imaging molecular charge states will help with the development of new materials and devices—and will improve our understanding of nature in general.
The study is published in the journal Science.