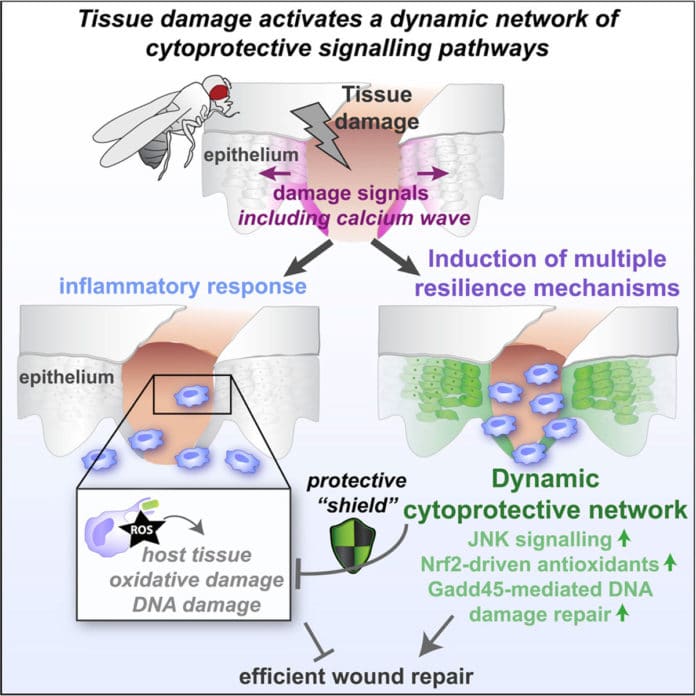
In healthy individuals, injured tissues rapidly repair themselves following damage. The body quickly recruits immune cells to the injury site where they fight infection by engulfing and killing invading pathogens, through the release of toxic factors such as unstable molecules containing oxygen known as “reactive oxygen species,” e.g., peroxides.
Incomprehensibly, while these exceptionally responsive ROS give protection from infection, they are additionally dangerous to host tissues and may eventually delay the repair. To counteract these destructive impacts, the repairing tissue activates powerful defensive machinery to ‘shield’ itself from the damage.
Now, scientists from the Bristol’s School of Biochemistry used in vivo time-lapse imaging and genetic manipulation techniques to map the exact identities of these protective pathways and identified how to stimulate this process in naïve tissues.
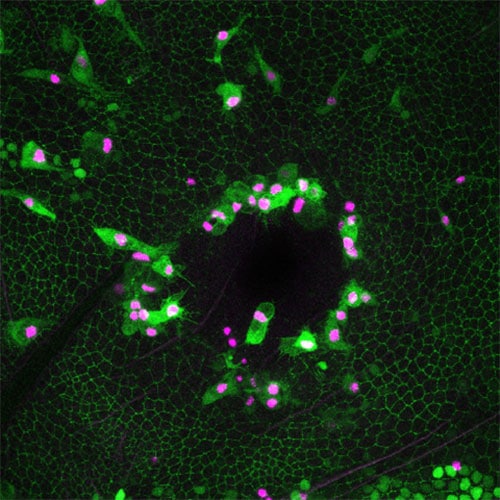
Dr. Helen Weavers from Bristol’s Faculty of Life Sciences and the study’s lead author explains: “In healthy individuals, injured tissues normally quickly repair themselves following damage. Within a healing skin wound, a stress-response is activated that recruits inflammatory cells, which in turn release a multitude of bacteriocidal factors, including reactive oxygen species (ROS), to eliminate invading pathogens.”
“In this study, we used translucent fruit flies to watch wound repair live as it happens and follow the behavior of the recruited immune cells. In doing so, we uncovered a network of protective pathways that shield tissues from inflammatory damage and make repairing tissues more ‘resilient’ to stress. We also demonstrated that ectopic activation of these pathways further enhanced tissue protection, whilst their inhibition led to significant delays in wound closure.”
“Now we know their identities, and how they are activated, we hope to develop ways to stimulate this protective machinery in patients prior to elective surgery.”
The discoveries have clear clinical relevance to patients because the therapeutic activation of these cytoprotective pathways in the clinic could also offer an energizing way to deal with ‘precondition’ patient tissues before elective surgery.
Dr. Weavers added: “We are now uncovering even more ‘resilience’ pathways that help to protect our body tissues from stress, both at sites of wounding and in other vulnerable organs that are often exposed to similar stressors. Since we find that the protection machinery is activated by the same pathways that also initiate the inflammatory response, we think the resilience machinery has evolved as a fail-safe mechanism for tissue protection each time inflammation is triggered.”
The study is published in the journal Current Biology.