Antibiotic resistance is a growing global concern that threatens the effective treatment of bacterial infections. The need for rapid and accurate methods to test for antibiotic resistance has become increasingly urgent. In recent years, researchers have developed a simple, fast, and affordable method for testing antibiotic resistance using optical nanomotion.
EPFL and Vrije Universiteit Brussel scientists have created a new and highly effective method for quickly testing antibiotic susceptibility. The technique, called Optical Nanomotion Detection, utilizes optical microscopy, is very fast, can detect single cells, requires no labeling, and only needs a basic optical microscope with a camera or even a mobile phone.
Optical nanomotion is a technique that utilizes light scattering to monitor the movements of small particles, including single bacteria. In this method, bacteria are immobilized on a surface and exposed to different antibiotics. By tracking the movements of the bacteria with a laser, the researchers can determine if the bacteria are alive or dead and if they are resistant or susceptible to antibiotics.
This optical nanomotion method is advantageous compared to traditional methods of antibiotic resistance testing, which can be time-consuming and expensive. The optical nanomotion method requires only a small amount of bacterial sample and can provide results within a matter of hours. In contrast, traditional methods can take days to produce results and require larger amounts of bacterial culture.
“We have developed a technique in our laboratories that allows us to obtain an antibiogram within 2-4 hours – instead of the current 24 hours for the most common germs and one month for tuberculosis,” says Dr. Sandor Kasas at EPFL. Professor Ronnie Willaert at Vrije Universiteit Brussel adds: “Our technique is not only faster but also simpler and much cheaper than all those existing now.”
Antibiotic resistance is a global public health issue responsible for millions of deaths yearly. Current antibiotic sensitivity testing methods are slow and require expensive equipment. However, researchers have now developed a simple, fast, and widely accessible method called optical nano motion detection (ONMD) that can perform an AST with single-cell sensitivity and without labeling bacteria.
The ONMD technique monitors the microscopic oscillations of bacterial cells before and while being exposed to antibiotics using a basic optical microscope, a camera, or a mobile phone. The technique successfully detected the sensitivity of various bacteria to antibiotics in under two hours, making it a simple and rapid way to assess bacterial cells’ sensitivity or resistance to antibiotics.
The authors state: “The simplicity and efficiency of the method make it a game-changer in the field of AST,” as it can be applied to a wide range of bacteria, which has significant implications for clinical and research applications.”
The rise of bacterial resistance to antibiotics is a severe public health concern. Rapid antibiotic susceptibility testing (AST) methods are needed to start the therapeutic process with the most efficient drug.
Traditional replication-based AST methods are slow, but some rapid AST techniques have been developed, which are complex and need sophisticated and costly equipment. Researchers have developed a rapid, label-free AST method called optical nano motion detection (ONMD), which monitors the microscopic oscillations of bacterial cells.
The technique only requires a basic traditional optical microscope, a camera/mobile phone, and dedicated software, and it can perform AST with single-cell sensitivity. The experiments successfully demonstrated that ONMD monitors life–death transitions and variations in the bacterial metabolic level induced by different nutrient concentrations.
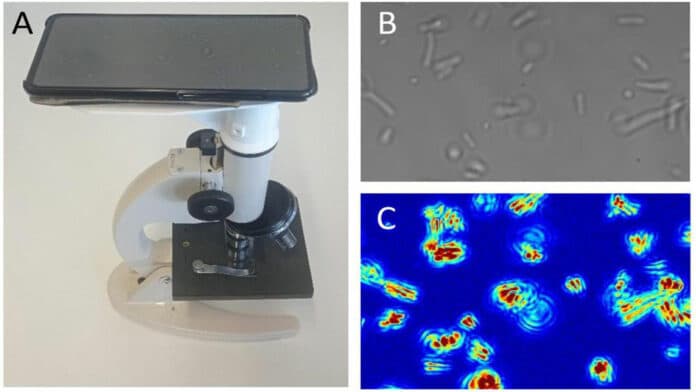
A very small drop of bacteria solution is put into a tiny space called a microfluidic analysis chamber. This chamber is then placed under a microscope, and the bacteria are observed for 10 seconds while a camera records their movement.
A special computer program called MATLAB is used to analyze the movement of each bacteria cell during 200 frames, which helps to understand how far they moved. To avoid the bacteria sticking to the chamber, they can be mixed in oil before the experiment. More information on how this is done can be found in a document called “SI Appendix.”
The scientists made a small chamber to put bacteria in, and they used a special camera to record how the bacteria moved around. They looked at how changing the environment affected the bacteria’s movements and found that the amount of nutrients and glucose had a significant impact. They also tested how different antibiotics affected the bacteria’s movements and found that some antibiotics greatly slowed them down. They compared the movements of living bacteria to those that were already dead.
The quick identification of the right antibiotic is important to prevent the spread of multi-resistant bacteria. This new technique is fast, easy to use, and can detect the movement of individual bacteria without needing any labels or attachments. It’s also much quicker and cheaper than other similar methods.
This technique works regardless of the bacteria’s growth rate, cell wall structure, or motility. It can be used to study the bacteria’s viability and assess their movement in different conditions, including antibiotic exposure. This technique has the potential to be used in developing countries for quick antibiotic testing. It can also be used to search for living organisms in extreme environments without relying on specific chemistry, such as on other planets.
Overall, the optical nanomotion method provides a fast, cheap, and easy way to test for antibiotic resistance in bacteria. As antibiotic resistance continues to be a significant health threat worldwide, this innovative method could play a critical role in developing new and effective antibiotic therapies.
Journal Reference:
- Maria I. Villalba, Eugenia Rossetti , Allan Bonvallat et al. Simple optical nano motion method for single-bacterium viability and antibiotic response testing. PNAS. DOI: 10.1073/pnas.2221284120