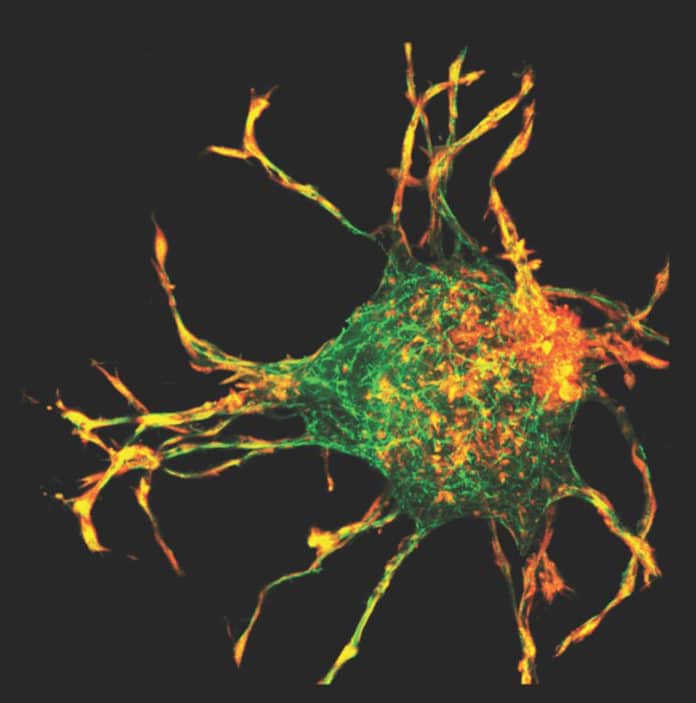
Wound healing is a complex and dynamic process of replacing devitalized and missing cellular structures and tissue layers. Now, a new study reveals how the process begins, which may provide new insight into wound healing, fibrosis, and cancer metastasis.
Scientists from the Washington University in St. Louis discovered the way fibroblasts, or common cells in connective tissue, interact with the extracellular matrix. This provides structural support as well as biochemical and biomechanical cues to cells.
Scientists revealed a recursive procedure that goes on between the cells and their environment as well as structures in the cells that were previously unknown.
Delaram Shakiba, a postdoctoral fellow from the NSF Science and Technology Center for Engineering Mechanobiology (CEMB) at the McKelvey School of Engineering, said, “Clinical efforts to prevent the progression of fiber contractile diseases, such as scarring and fibrosis, have been largely unsuccessful, in part because the mechanisms that cells use to interact with the protein fibers around them are unclear. We found that fibroblasts use completely different mechanisms in the early — and I think the most treatable — stages of these interactions and that their responses to drugs can, therefore, be the opposite of what they would be in the later stages.”
“The process has stymied mechanobiology researchers for some time.”
“Scientists in the field of mechanobiology thought that cells pulled in collagen from the extracellular matrix by reaching out with long protrusions, grabbing it and pulling it back. We discovered that this wasn’t the case. A cell has to push its way out through collagen first, then instead of grabbing on, it essentially shoots tiny hairs, or filopodia, out of the sides of its arms, pulls in collagen that way, then retracts.”
Next, in collaboration with scientists at CEMB at the University of Pennsylvania, scientists were able to validate some mathematical models to go through the engineering process. Interestingly, they now have the basic rules that cells follow. They can now begin to design specific stimuli to direct a cell to behave in a certain way in building a tissue-engineered structure.
Scientists learned they could control the cell shape in two ways: First, by controlling the boundaries around it, and second, by inhibiting or upregulating particular proteins involved in the remodeling of the collagen.
Fibroblasts pull the edges of a wound together, causing it to contract or close up. Collagen in the cells then remodels the extracellular matrix to close the wound fully. This is where mechanobiology comes into play.
Genin said, “There’s a balance between tension and compression inside a cell that is newly exposed to fibrous proteins. There is tension in actin cables, and by playing with that balance, we can make these protrusions grow extremely long. We can stop the remodeling from occurring, or we can increase it.”
By using a 3D-mapping technique with a computational model, scientists calculated the 3D strain and stress fields created by the protrusions from the cells. As cells accumulated collagen, tension- driven redesigning and alignment of collagen fibers prompted the development of collagen tracts. This requires helpful cooperation among cells, through which cells can connect precisely.
Elliot Elson, professor emeritus of biochemistry and molecular biophysics at the School of Medicine, said, “New methods of microscopy, tissue engineering, and biomechanical modeling greatly enhance our understanding of the mechanisms by which cells modify and repair the tissues they populate. Fibrous cellular structures generate and guide forces that compress and reorient their extracellular fibrous environment. This raises new questions about the molecular mechanisms of these functions and how cells regulate the forces they exert and how they govern the extent of matrix deformation.”
Genin said, “Wound healing is a great example of how these processes are important in a physiologic way. We’ll be able to come up with insight in how to train cells, not to excessively compact the collagen around them.”
Journal Reference:
- Delaram Shakiba et al. The Balance between Actomyosin Contractility and Microtubule Polymerization Regulates Hierarchical Protrusions That Govern Efficient Fibroblast–Collagen Interactions. ACS Nano, 2020; DOI: 10.1021/acsnano.9b09941