Scientists at the Department of Chemistry at Imperial College London have demonstrated a new way to predict the outcome of reactions. They used a computer-driven approach to predict the results of cage-building reactions with high precision.
In every chemical and biological process, enzymes have a significant role. The enzymes take biomolecules into cavities within their structures and facilitate essential reactions.
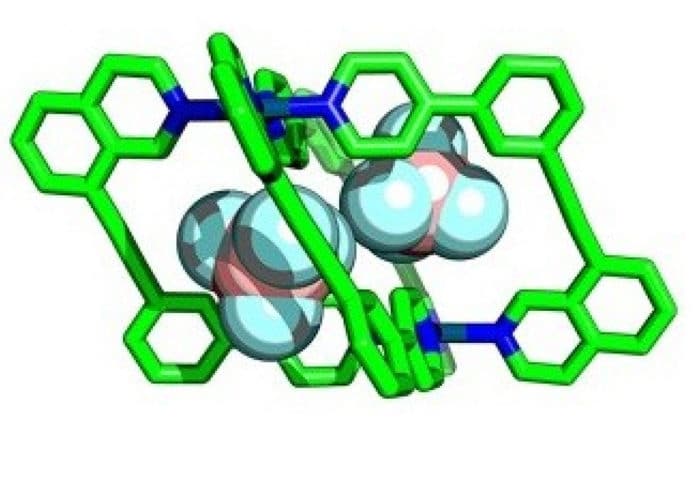
Chemists have attempted to emulate this with ‘molecular cages’ – chemical structures that contain cavities that can bind smaller molecules within, called ‘guests. These molecular cages can act as artificial enzyme-mimics. They also have the potential to accelerate essential reactions.
Improvements in such reactions could lead to the development of new technologies in areas such as chemical sensing. Although, designing such structures can be a challenging task for scientists.
This new study has addressed that issue: a new approach to predict the results of cage-building reactions with high precision. The approach can help scientists select perfect building blocks to prepare cages with desirable structures and properties.
Simplifying the synthesis of molecular cages requires most cages and their cavities to be highly symmetric. Although, this limits the design of cages for potential guest molecules. This contrasts with natural enzymes’ ability to be highly selective in which molecules can bind with them.
By using more complex components in their construction, scientists are developing ways to assemble cages with lower symmetry, allowing more guest-specific cavity shapes. Using unsymmetrical building blocks, scientists can create wonky’ cages with interesting cavity shapes.
However, creating unsymmetrical building blocks is difficult as the outcomes of the ‘self-assembly’ reactions needed to construct them are harder to predict. Failed reactions can result in the formation of an undesirable molecule, or even a mixture of products, rather than the single target structure.
The new approach instead analyses computational models of potential cages to make predictions about their self-assembly. The predictions use the energy and geometry of the computationally constructed cages and are good guides for whether a self-assembly process will lead to a single structure. This information can then be used to select target cage molecules to prepare in the lab.
Study co-author Dr. Jamie Lewis from the Department of Chemistry at Imperial said: “Previously, we’ve just had to get in the lab and try lots of things until something worked. Now we can run some quick calculations, identify cages with useful properties, and be confident that we can synthesize them without any problems.”
Lead author of the study Dr. Andrew Tarzia, from the Department of Chemistry at Imperial, said: “The efficiency of our approach is the key because it allows us to test on a computer many more building blocks in a week than could be tested in the laboratory and with more diversity too.”
Using the approach, scientists were able to predict the experimental outcomes of the self-assembly process successfully. They also created several new low-symmetry ‘wonky cages’ that had never been synthesized before.
The team are now continuing to develop and improve this approach to computationally informed, efficient synthesis to access new molecular cages.
Journal Reference:
- Andrew Tarzia, James E. M. Lewis et al. High-throughput Computational Evaluation of Low Symmetry Pd2L4 Cages to Aid in System Design. DOI: 10.1002/anie.202106721