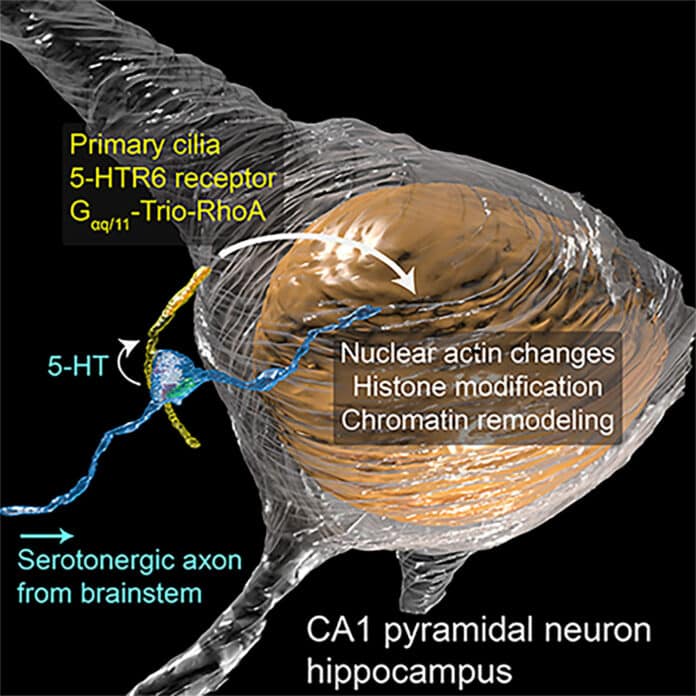
Synapses refer to the contact points between neurons where information is passed from one neuron to the next. They usually occur between the axon of one neuron and the dendrites of other neurons. Until now, synapses were never observed between the neuron’s axon and the primary cilium.
Using high-resolution microscopes and innovative tools, scientists at HHMI’s Janelia Research Campus have peer deep into the cell and cilia to observe the synapse. They discovered a new kind of synapse in the tiny hairs on the surface of neurons. This particular synapse represents a way to change what is being transcribed or made in the nucleus, which changes whole programs.
Janelia Senior Group Leader David Clapham, whose team led the new research, said, “The effects in the cell are not just short-term, some can be long-term. It is like a new dock on a cell that gives express access to chromatin changes, which is very important because chromatin changes so many aspects of the cell.”
The Discovery of this new kind of synapse could help scientists better understand how long-term cell changes are communicated. The cilia, which extend from the inside of the cell, close to the nucleus, to the surface, may offer a quicker and more focused method for cells to carry out these long-term alterations.
Cilia- tiny hair-like organelles attached to the cell surface- play an essential role in cell division during development. However, it remains elusive why other cells in our bodies, including neurons, retained this hair-like, bacterium-sized protrusion into maturity.
Due to the fact that these cilia were challenging to see using conventional imaging methods, scientists have generally overlooked them. However, more recent advancements in imaging technology have generated interest in these tiny appendages.
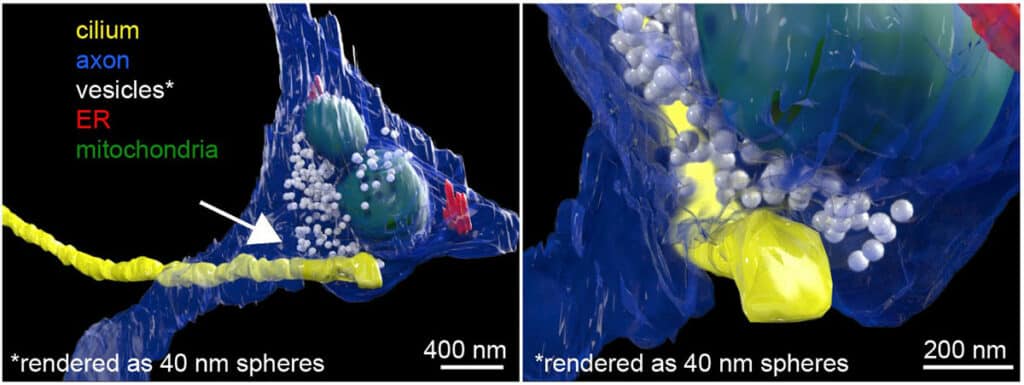
Using focused ion beam-scanning electron microscopy, or FIB-SEM, scientists take a look at the cilia. They observed a connection, or synapse, between the neuron’s axon and the cilium protruding outside the cell body. The team refers to these connections as “axon-cilium” or “axo-ciliary” synapses because of their structural similarities to those of known synapses.
Later, scientists developed new biosensors and chemical tools to determine the Function of this new synapse. They used fluorescence lifetime imaging (FLIM) to better measurements of biochemical events inside the cilia.
Shu-Hsien Sheu, a senior scientist at Janelia and the first author of the new study, said, “I learned FLIM during the pandemic to address some of the technical challenges. It turned out to be a game changer.”
Using these tools, scientists showed stepwise- how the neurotransmitter serotonin is released from the axon onto receptors on the cilia. This triggers a signaling cascade that opens the chromatin structure and allows changes to genomic material in the cell’s nucleus.
Sheu said, “Function is what makes static structures come alive. Once confident about the structural finding, we looked deeply into its functional properties.”
Sheu says, “HHMI’s curiosity-driven research philosophy enabled the Discovery, which may not have been possible in a traditional research setting. This is a good example of how we can make observations into discoveries.”
Scientists noted, “Because the signals passed across the ciliary synapse enable changes to genomic material in the nucleus, they are likely responsible for longer-term changes in neurons than signals passed from axons to dendrites. These changes could last from hours to days to years, depending on the chromatin encodes proteins.”
Scientists mainly observed receptors for serotonin. There are at least seven to 10 other receptors on cilia for different neurotransmitters that will now need to be examined. Cilia on other cells beyond the brain, like the liver and kidney, also deserve a closer look.
A better understanding of the role of these ciliary synapses and receptors could help scientists develop more selective medications. Drugs that target serotonin transporters are used to treat depression, while serotonin is also linked to our sleep-wake cycle.
Journal Reference:
- Shu-Hsien Sheu, Srigokul Upadhyayula, Vincent Dupuy, et al. A serotonergic axon-cilium synapse drives nuclear signaling to alter chromatin accessibility. Cell. DOI: 10.1016/j.cell.2022.07.026