Neonatal seizures or neonatal convulsions are epileptic fits occurring from birth to the end of the neonatal period. The neonatal period is the most vulnerable of all periods of life for developing seizures, particularly in the first 1–2 days to the first week from birth.
Neonatal seizures can be difficult to diagnose because the seizure may be short and subtle. Thus, they sometimes signify serious malfunction of or damage to the immature brain and constitute a neurological emergency demanding urgent diagnosis and management.
Scientists at the Penn State University now have come up with a new drug that could proficiently treat neonatal seizures in rodent models.
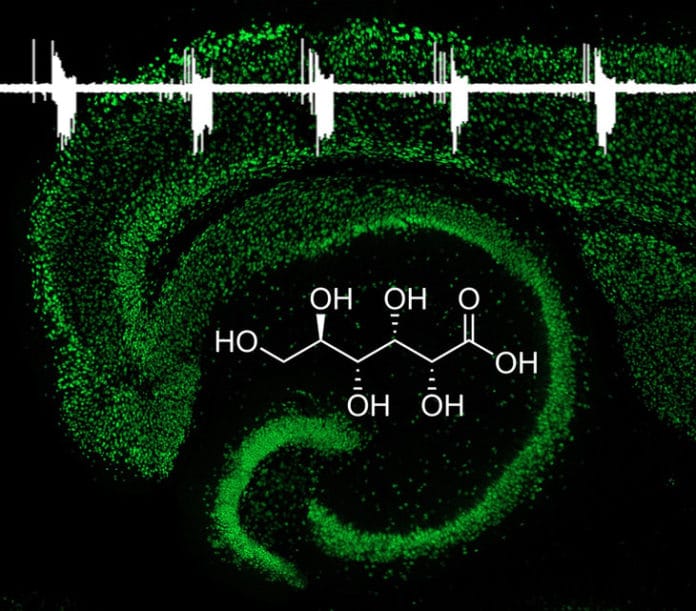
They developed this new drug by identifying gluconate that acts as an anti-convulsant and inhibits seizures by targeting the activity of channels that control the flow of chloride ions in and out of neonatal neurons.
Gong Chen, professor of biology said, “Neonates are the most vulnerable population to seizures, but there is still no effective medication for the treatment of neonatal epilepsy. The incidence of epilepsy is highest in the first year of life, with two to four infant babies suffering from neonatal epilepsy for every 1,000 live births in the United States. Unfortunately, so far there is no effective drug available that is specifically developed for neonatal epilepsy patients.”
Neonatal epilepsy patients are often resistant to or do not respond to current anti-epilepsy drugs, and long-term use of some of these treatments may have side effects on brain development.
In this study, scientists demonstrated that gluconate can inhibit seizure activity in neonatal neurons. More importantly, gluconate suppresses seizure activity in neonatal animals more effectively than in adults.
Gluconate inhibits neonatal seizures by targeting what are known as CLC-3 chloride channels. These channels mediate a large ion current in neonatal brains but are less active in adult brains. Gluconate appears to be too large to pass through the small openings of the CLC-3 channels and therefore acts as a channel blocker.
Chen said, “This is truly exciting because we have finally identified a potential anti-convulsant drug that shows preference to inhibit neonatal seizure activity.”
Zheng Wu, an assistant research professor at Penn State and the first author of the paper said, “Gluconate is a small organic compound that is produced through the oxidization of glucose in many plants, fruits and honey. It has minimal side effects when compared to other organic ions. Because of this, our discovery of its anti-seizure function in neonates could have an accelerated path toward therapeutic development for use in the treatment of neonatal epilepsy.”
“We were surprised that gluconate targeted CLC-3 chloride channels because they have been associated with regulating neuronal transmission, glioma proliferation, and neuron cell death, but until now, there was no mention of neonatal seizure at all. Here we found not only that CLC-3 chloride channels are highly expressed in the neonatal brain but also that they are closely related to neonatal seizures. Importantly, gluconate not only blocks the CLC-3 chloride channels but it also significantly inhibits neonatal seizure activity. Its neonatal activity makes it a great specific target for neonatal anti-epilepsy drugs.”
In addition to gluconate, a ketone body called β-HB that generated in the liver under a ketogenic diet can also act as an inhibitor of CLC-3 chloride channels and suppress neonatal seizure activity.
Chen said, “The similar effects of gluconate and a ketone body in inhibiting both CLC-3 chloride channels and neonatal seizures may suggest a better treatment than a ketogenic diet in suppressing neonatal seizures. Our studies not only identify a new drug target, the CLC-3 chloride channels, for neonatal epilepsy but also discovered two potential anticonvulsant drugs, gluconate and the ketone body β-HB, that can suppress neonatal seizures. Our work also opens a new avenue for other scientists to design even more specific and potent blockers for CLC-3 chloride channels to treat neonatal epilepsy.”
Other co-authors of the study include Fengping Dong, Mengyang Feng, Yue Wang, Yuting Bai, Gangyi Wu, and Bernhard Lüscher at Penn State; Qingwei Huo and Cheng Long at South China Normal University; Liang Ren and Yun Wang at Fudan University, Sheng-Tian Li at Shanghai Jiao Tong University; and Guan-Lei Wang at Sun Yat-sen University.