The long charging time of electric vehicles (EVs) compared with the refueling time of gasoline vehicles has been a major barrier to the mass adoption of EVs. For a recharging experience comparable to that of gasoline vehicles, the US Department of Energy (DOE) has set a Fast-Charge goal of less than 15 minutes to charge an EV battery.
However, fast-charging current Li-ion batteries can lead to Li-metal plating of the carbon anode and the potential formation of catastrophic lithium dendrite shorts. This is where Li-metal anodes come in, as they have the potential to overcome these issues. Li-metal anodes allow for higher energy density batteries and thus longer EV range. However, the charging rate of Li-metal anodes has been limited so far due to the formation of lithium dendrite shorts.
To address this, a research team from the Maryland Energy Innovation Institute (MEI2) at the University of Maryland (UMD) has developed a single-phase mixed ion- and electron-conducting (MIEC) garnet material with comparable Li-ion and electronic conductivities.
When integrated into their previously developed 3D architecture, the material not only achieved the DOE Fast-charge goal for Li cycling but exceeded it by a factor of 10.
The porous structure of the MIEC garnet can be quite helpful in relieving the stresses on solid electrolytes during cycling. By spreading the potential uniformly across the surface, it can help prevent local hot spots that might otherwise induce the formation of dendrites.
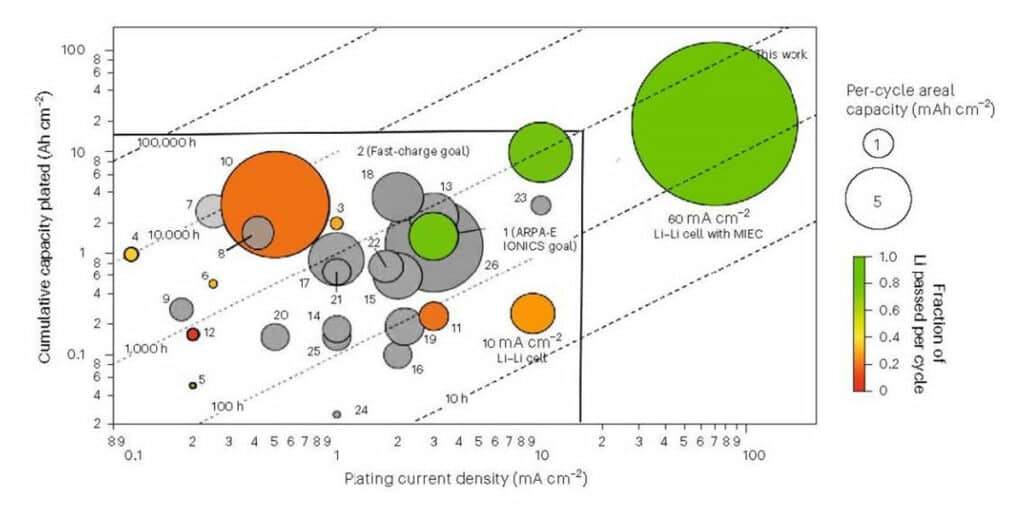
As the above image shows, the Li cycling rates, quantity of Li per cycle (circle diameter), and cumulative Li cycling far exceed the DOE Fast Charge Goals for current density, per-cycle areal capacity, and cumulative capacity at room temperature with no applied pressure.
Researchers demonstrated that in a trilayer architecture with a porous MIEC framework supporting a thin, dense garnet electrolyte, the critical current density could be increased to a previously unheard-of 100 mA cm−2 with no dendrite-shorting. In addition, it was shown that symmetric Li cells could be continuously cycled at a current density of 60 mA cm−2 with a maximum per-cycle Li plating and stripping capacity of 30 mAh cm−2, which is six times the capacity of state-of-the-art cathodes.
Moreover, a cumulative Li plating capacity of 18.5 Ah cm−2 was achieved with the transformative architecture. If paired with a state-of-the-art cathode areal capacity of 5 mAh cm−2, the structure would yield a projected 3,700 cycles. With this Li cycling capability, EVs could do 100% depth of discharge cycles every day for ten years, significantly surpassing requirements for commercial electric vehicle battery lifetimes.
“Dr. Eric Wachsman [lead researcher] and team demonstrated superior rate capability of lithium metal anode in this work; it is through innovative 3D design and the unique architecture such performance could be achieved. Such an approach opens up a new paradigm for the design of next-generation high-energy rechargeable batteries,” said Dr. Y. Shirley Meng, Chief Scientist at the University of Chicago.
The successful demonstration of this high-rate dendrite-free Li metal in 3D MIEC structures is expected to spur the development of practical solid-state batteries with Li-free anodes.
Journal reference:
- George V. Alexander, Changmin Shi, Jon O’Neill & Eric D. Wachsman. Extreme lithium-metal cycling enabled by a mixed ion- and electron-conducting garnet three-dimensional architecture. Nature Materials, 2023; DOI: 10.1038/s41563-023-01627-9
