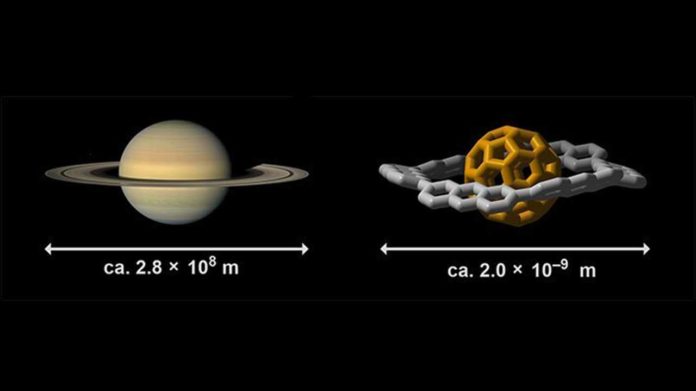
Saturn, the second biggest planet in our solar system, has characteristic rings that are held in orbit by gravitational forces. These days, nanoscale molecular complexes looking like a small form of Saturn are being explored for their potential applications in future materials.
A group of analysts at Tokyo Tech in a joint effort with Okayama University of Science, Japan, has given exploratory confirmation demonstrating the development of a specific nano-Saturn system.
Scientists showed that nanomolecular structures composed of hydrogen and carbon can take the form of a combined ring and sphere, which looks like the planet Saturn. They analyzed the interactions between the ring and the sphere and explained their origin.
According to scientists, this discovery may have implications for the development of new carbon nanomaterials in the future.
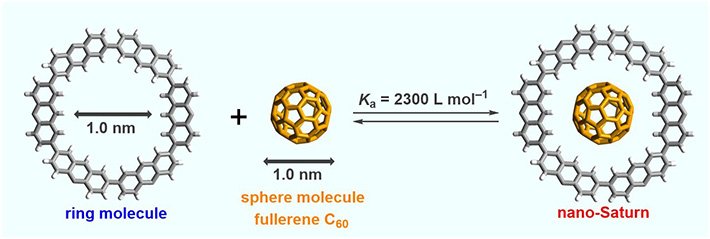
Nano-Saturn systems are composed of a spherical molecule and a ring-like molecule. Unlike the rings of Saturn, which are held close to the planet by gravity, the ring molecule hosts the spherical molecule inside itself. While previous studies resulted in systems with similar structures, the ring molecules used in those complexes resembled more of a belt or a tire wrapped around a sphere instead of an almost flat disk. This is important because the shape of the molecules used influences the type of bonding shown by the host and the guest molecules, which in turn determines the material properties.
For their nano-scale Saturn-like system, the group utilized an extremely understood sort of carbon particle called fullerene (C60) as the “planet.” C60 is made of carbon shapes looking like pentagons and hexagons that bond together to form a hollow sphere, resembling the leather patches in soccer balls.
Some of these electrons in fullerene are called π-electrons and shape an “electron cloud” that can move and bind with other molecules, for example, the ones utilized as the “ring,” which additionally has π-electrons and carbon-hydrogen (CH) groups.
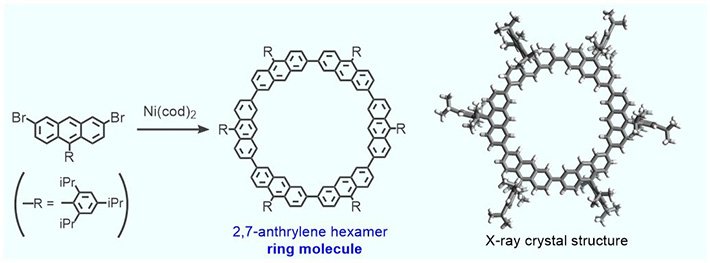
The ring-like molecule utilized by the group is called cyclic 2,7-anthrylene hexamer, and is created. The diameter of the subsequent internal cavity is relatively indistinguishable to the diameter of the fullerene sphere, empowering the molecules to indicate CH···π connections. Along these lines, the nano-Saturn framework is a potential model for researching this sort of cooperation amongst C60 and arene molecules.
X-ray analyses, nuclear magnetic resonance (NMR) measurements, and density functional theory (DFT) calculations showed that the nano-Saturn system as a whole is more stable than the individual host and guest molecules not only in the solid state but also in solution. The association constant for the formation of the nano-Saturn system was determined to be 2,300 L mol–1, being 4 times larger than that of a previous nano-Saturn system using a flat-shaped outer ring.
Shinji Toyota, research led said, “the formation of this Saturn-type complex and other host-guest systems should be further studied to develop innovative applications of these complexes for functional materials in the future.”
Scientists published their research in the journal Angewandte Chemie International Edition.