Scientists at Stanford University, in a recent study, have suggested that converting methane to CO2 may help combat climate change. Doing this could help turn the tide of climate change while also turning a healthy profit. According to scientists, swapping methane for carbon dioxide is a significant net benefit for the climate.
In 2018, methane — about 60 percent of which is generated by humans — reached atmospheric concentrations two and a half times greater than pre-industrial levels, previous studies have shown.
Although the amount of carbon dioxide in the atmosphere is much greater, methane is 84 times more potent in terms of warming the climate system over the first 20 years after its release.
According to the researchers, the basic idea is that some sources of methane emissions — from rice cultivation or cattle, for example — may be very difficult or expensive to eliminate.
Chris Field, the Perry L. McCarty Director of the Stanford Woods Institute for the Environment said, “The basic idea is that some sources of methane emissions – from rice cultivation or cattle, for example – may be very difficult or expensive to eliminate. An alternative is to offset these emissions via methane removal, so there is no net effect on warming the atmosphere.”
Most scenarios for removing carbon dioxide typically assume hundreds of billions of tons removed over decades and do not restore the atmosphere to pre-industrial levels. In contrast, methane concentrations could be restored to pre-industrial levels by removing about 3.2 billion tons of the gas from the atmosphere and converting it into an amount of carbon dioxide equivalent to a few months of global industrial emissions, according to the researchers. If successful, the approach would eliminate approximately one-sixth of all causes of global warming to date.
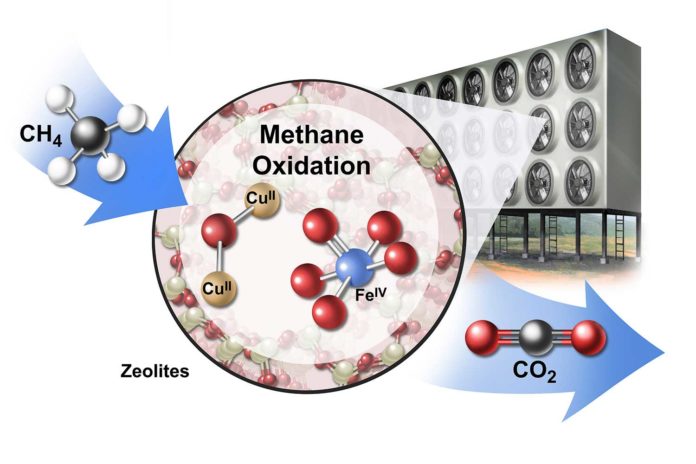
Methane is challenging to capture from the air because its concentration is very low. However, the scientists noted that zeolite, a crystalline material that consists primarily of aluminum, silicon, and oxygen, could act essentially like a sponge to soak up methane.
Ed Solomon, the Monroe E. Spaght Professor of Chemistry in the School of Humanities and Sciences said, “The porous molecular structure, relatively large surface area and ability to host copper and iron in zeolites make them promising catalysts for capturing methane and other gases.”
Scientists noted, “The whole process might take the form of a giant contraption with electric fans forcing air through tumbling chambers or reactors full of powdered or pelletized zeolites and other catalysts. The trapped methane could then be heated to form and release carbon dioxide.”
The study, published in Nature Sustainability on May 20.