Cartilage is a strong, flexible connective tissue in joints that acts like a cushion to reduce friction and prevent bones from rubbing together when you use your joints. It is essential for healthy movement but has almost no regenerative ability in adults. As a result, damage to cartilage due to injury or wear-and-tear can lead to painful chronic conditions like osteoarthritis.
While scientists are exploring the possibility of creating artificial replacement materials, it seems that nothing can beat the performance of natural cartilage. Another potential solution is to use stem cells to regenerate natural cartilage, but this poses other challenges, such as getting the stem cells to grow in the right shape, as clumps of these cells often change shape or shrink.
Now, TU Wien (Vienna) researchers have developed a new way to grow cartilage from stem cells.
Using a novel high-resolution 3D printing technique, researchers have created tiny cage-like structures that look like mini footballs and have a diameter of just a third of a millimeter. These spheroids can be arranged in any geometry and can be connected to each other like building blocks, providing a scaffold for the cartilage stem cells inside.
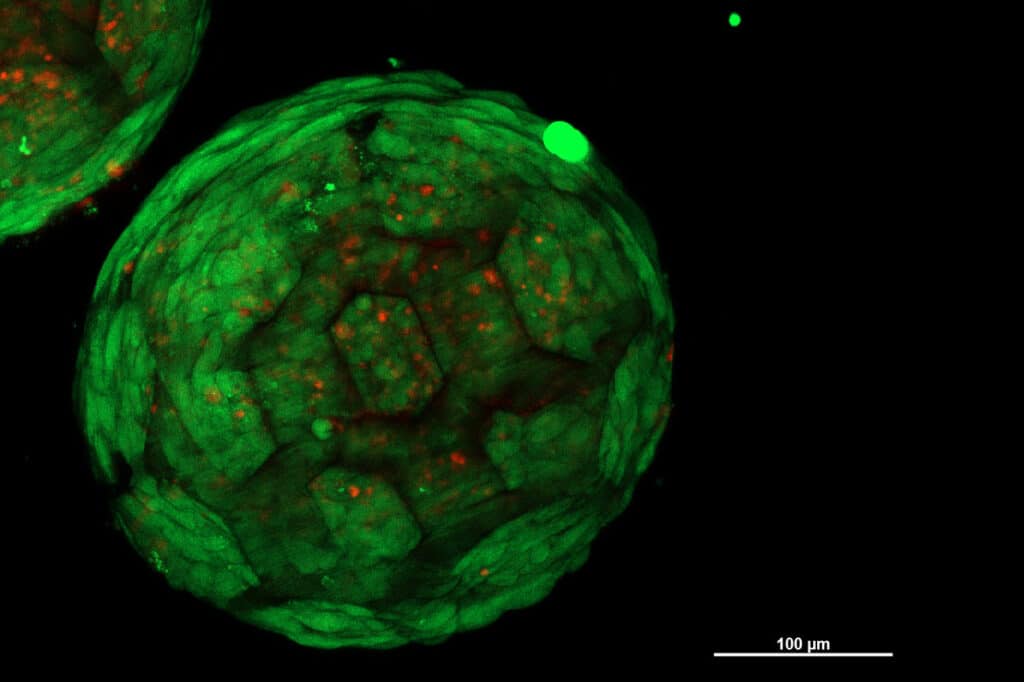
The researchers have demonstrated this approach with cartilage tissue, which was previously considered particularly challenging in this respect.
“Under the microscope, you can see very clearly: neighboring spheroids grow together, the cells migrate from one spheroid to the other and vice versa, they connect seamlessly and result in a closed structure without any cavities – in contrast to other methods that have been used so far, in which visible interfaces remain between neighboring cell clumps,” said Oliver Kopinski-Grünwald, an author of the study.
The 3D-printed porous spheroids are made of biocompatible and degradable plastic, which provides stability and structure initially but breaks down over time, leaving behind the required tissue shape. These spheroids can then be arranged in any geometry, and the cells of the different units combine seamlessly to form a uniform, living tissue. Researchers say this could be a game changer for cartilage replacements, allowing for more effective and customizable solutions.

“An initial goal would be to produce small, tailor-made pieces of cartilage tissue that can be inserted into existing cartilage material after an injury,” says Oliver Kopinski-Grünwald in a statement. “In any case, we have now been able to show that our method for producing cartilage tissue using spherical micro-scaffolds works in principle and has decisive advantages over other technologies.”
The new approach has great potential beyond cartilage tissue and could be applied to larger tissues such as bone tissue. However, there are still some challenges to overcome, especially when it comes to incorporating blood vessels for larger tissues.
Journal reference:
- Oliver Kopinski-Grünwald, Olivier Guillaume, Tamara Ferner, Barbara Schädl, Aleksandr Ovsianikov. Scaffolded spheroids as building blocks for bottom-up cartilage tissue engineering show enhanced bioassembly dynamics. Acta Biomaterialia, 2024; DOI: 10.1016/j.actbio.2023.12.001