There are currently no treatments for geographic atrophy, the advanced form of age-related macular degeneration. Hence, innovative studies are needed to model this condition and prevent or delay its progression.
The discovery of the disease’s genetic signatures could lead to better diagnosis and treatment of age-related macular degeneration. By reprogramming stem cells, scientists- from the Garvan Institute of Medical Research, the University of Melbourne, the Menzies Institute for Medical Research at the University of Tasmania, and the Centre for Eye Research Australia- created models of diseased eye cells. They then analyzed DNA, RNA, and proteins to pinpoint the genetic clues.
The team examined the effects of genetic variations on the cells causing age-related macular degeneration. To identify the genetic indicators of this disease at the smallest scale possible, they focused on particular cell types.
Joint lead author Professor Joseph Powell, Pillar Director of Cellular Science at Garvan, said, “This is the basis of precision medicine, where we can then look at what therapeutics might be most effective for a person’s genetic profile of the disease.”
Scientists took skin samples from 79 participants with and without the late stage of AMD, called geographic atrophy. Their skin cells were reprogrammed to become induced pluripotent stem cells, which were subsequently directed by molecular cues to become retinal pigment epithelium cells, the cells impacted by AMD.
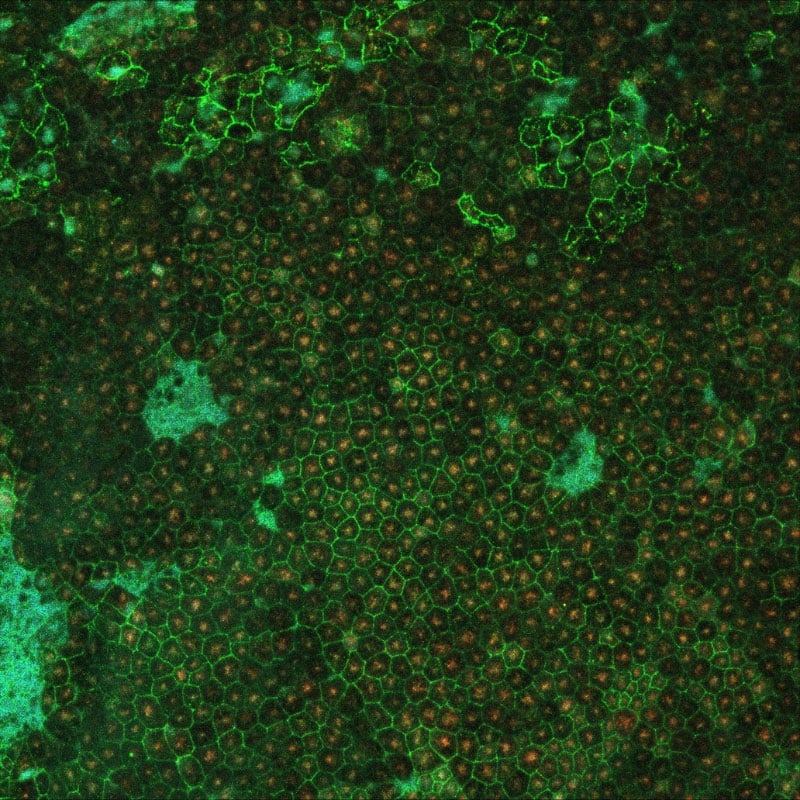
The retina’s back is lined with cells called retinal pigment epithelium, which is crucial to the retina‘s health and functionality. The loss of vision in AMD is brought on by the degeneration of photoreceptors, which are light-sensing neurons in the retina that relay visual information to the brain.
An analysis of 127,600 cells revealed 439 molecular signatures associated with AMD, with 43 potential new gene variants. Key identified pathways were subsequently tested within the cells. They revealed differences in the energy-making mitochondria between healthy and AMD cells, rendering mitochondrial proteins potential targets to prevent or alter the course of AMD.
Further, the molecular signatures can now be used for screening treatments using patient-specific cells in a dish.
Co-lead of the study Professor Alice Pébay from the University of Melbourne, said, “Ultimately, we are interested in matching the genetic profile of a patient to the best drug for that patient. We must test how they work in cells relevant to the disease.”
In another recent study, scientists uncovered genetic signatures of glaucoma – a degenerative eye disease causing blindness – using stem cell models of the retina and optic nerve.
Journal Reference:
- Senabouth, A., Daniszewski, M., Lidgerwood, G.E. et al. Transcriptomic and proteomic retinal pigment epithelium signatures of age-related macular degeneration. Nat Commun 13, 4233 (2022). DOI: 10.1038/s41467-022-31707-4
