Bats are one of the essential natural reservoirs for various emerging viruses that induce severe illness, including severe acute respiratory syndrome coronavirus (SARS-CoV), Middle East respiratory syndrome coronavirus (MERS-CoV), Hendra virus, Ebola virus, Marburg virus, and SARS-CoV-2.
A single bat can host these viruses without getting sick; why bats can live with so many viruses without themselves falling ill remains one of the great mysteries of virology and its neighboring disciplines. And solving this mystery has been made all the more urgent by the ongoing COVID-19 pandemic.
Many experiments attempt to explain why bats can be infected with many viruses at a time without succumbing to diseases. This knowledge could help us reduce the threat to humans of infectious diseases we have struggled with to date.
Researchers have struggled until now with the fact that live wild bats make poor research subjects. To overcome this obstacle, for the first time, researchers have grown rousette bat “organoids,” which reproduce intestines in vitro.
A recent study, appearing in the International Journal of Molecular Sciences on October 5, described the bat organoid growth technique.
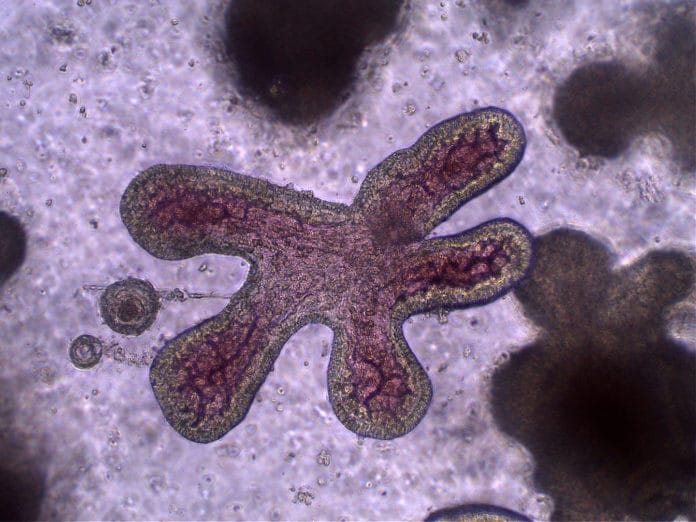
Researchers successfully developed intestinal organoids from a Megachiroptera, Rousettus leschenaultia, and established a long-term stable culture method for the first time. The rousette bat intestinal organoids showed the same cellular composition as intestinal tissues from morphological and immunohistochemical characterizing analyses, as demonstrated previously in bats and humans.
However, bats are wild animals, not experimental animals. It is more challenging to conduct reproducible animal experiments on bats than on more common experimental animals such as mice or pigs. Hence, most experiments have had to take place on cell lines taken from bats rather than bats themselves or bat organs.
“If this experimental blockage could be overcome, virus-bat relationships could be understood and lead to reduce human illness and death.,” said Tsutomu Omatsu, one of the authors of the paper and an associate professor with the Center for Infectious Diseases of Epidemiology and Prevention Research at Tokyo University of Agriculture and Technology.
They chose Rousette bats, a type of megabat or fruit bat because they are thought to be a natural reservoir of the filovirus family of viruses, including the Ebola and Marburg viruses. This particular species of rousette bat was also selected because, in previous research, another species of rousette bat, Rousettus leschenaultii, had shown a transient but not robust infection from an experimental inoculation of SARS-CoV-2. This virus causes COVID-19, while a cell line from the intestine of Rousettus leschenaultia had not been infected at all. Several species of flying foxes in Southeast Asia and Australia have also been found to be hosts of Pteropine orthoreovirus (PRV), which has caused respiratory disease in humans.
The researchers first had to find an optimum medium for the growth of bat intestine cells. They did this by culture organoids with nine different growth supplements (nutrients and other molecules that encourage cell proliferation). Three out of the nine achieved significantly higher cell growth and proliferation rates after seven days.
In addition, the rousette bat intestinal organoids grown with these three supplements were long-lived, maintaining active proliferation for up to ten passages (up to 10 times of reconstructions of organoids from separated cells that were composed of the previous organoids). Organoids that were long-term cryopreserved (frozen) could also grow normally once thawed.
The researchers deployed two techniques to confirm that the organoid mimics the epithelial (outer) tissue of the bat’s intestine—the part of the bat organ that first encounters virus particles and is thus of particular scientific interest. First, they used transmission electron microscopy to investigate the organoids’ cellular anatomy (histology). Second, they used immunofluorescence staining—a common method used to detect and visualize molecules in biological samples—to look for molecular markers that indicate that the tissue under investigation comes from bat intestines. Together, these two techniques told the researchers that the organoids were recreating the typical cellular components of rousette bat intestine tissues.
“The organoid showed susceptibility to Pteropine orthoreovirus (PRV) but not to SARS-CoV-2 in experimental inoculation. This is the first report of the establishment of an expandable organoid culture system of the rousette bat intestinal organoid and its sensitivity to bat-associated viruses, PRV and SARS-CoV-2. This organoid is a useful tool for the elucidation of tolerance mechanisms of the emerging rousette bat-associated viruses such as Ebola and Marburg virus.” Study quotes.
Having successfully produced bat organoids for the first time, the researchers now want to repeat their trick with other flying fox organs such as lungs, liver, and kidneys. The researchers will then infect this mass of bat ‘insides’ with highly pathogenic viruses to analyze their gene expression (turning genes on and off) in detail to clarify the mechanism of why bats can host such pathogens without getting sick.
Journal Reference
- Mohamed Elbadawy, Yuki Kato, Nagisa Saito, Kimika Hayashi, Amira Abugomaa, Mio Kobayashi, Toshinori Yoshida, Makoto Shibutani, Masahiro Kaneda, Hideyuki Yamawaki, Tetsuya Mizutani, Chang-Kweng Lim, Masayuki Saijo, Kazuaki Sasaki, Tatsuya Usui, and Tsutomu Omatsu. “Establishment of Intestinal Organoid from Rousettus leschenaultii and the Susceptibility to Bat-Associated Viruses, SARS-CoV-2 and Pteropine Orthoreovirus” International Journal of Molecular Sciences 22, no. 19: 10763, 2021. DOI: 10.3390/ijms221910763