A team of scientists has made the strongest silver that is 42 percent stronger than the previous world record.
What’s more, scientists have discovered a mechanism that enabled them to work at the nanoscale and make metals that are much stronger than anything ever made before without losing electrical conductivity.
This principal leap forward guarantees another classification of materials that can conquer a conventional exchange off in industrial and commercial materials between strength and ability to convey electrical flow.
Defects in metals sometimes lead to undesirable qualities, like brittleness or softening. This encourages scientists to create various alloys or heavy mixtures of material to make them stronger. But creating stronger material may cause the material to lose its conductivity.
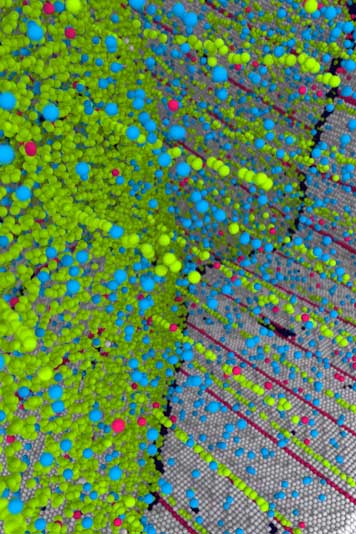
With an idea of mixing a trace amount of copper into the silver, scientists demonstrated it could change two sorts of inherent nanoscale defects into an incredible inside structure.
Frederic Sansoz, a materials scientist and mechanical engineering professor at the University of Vermont, said, “That’s because impurities are directly attracted to these defects. In other words, the team used a copper impurity—a form of doping or “micro-alloy” as the scientists style it—to control the behavior of defects in silver. Like a kind of atomic-scale jiu-jitsu, the scientists flipped the defects to their advantage, using them to both strengthen the metal and maintain its electrical conductivity.”
Scientists started with a foundational idea of materials engineering: as the size of a crystal—or grain—of material gets smaller, it gets stronger. Scientists call this the Hall-Petch relation. This general design rule has enabled researchers and specialists to build stronger alloys and advanced ceramics production for more than 70 years. It works well, overall.
Until it doesn’t. Eventually, when grains of metal reach an infinitesimally tiny size—under tens of nanometers wide—the boundaries between the grains become unstable and begin to move. Therefore, another known approach to strengthening metals like silver uses nanoscale “coherent twin boundaries,” which are a special type of grain boundary.
These structures of paired atoms—forming a symmetrical mirror-like crystalline interface—are exceedingly strong to deformation. Except that these twin boundaries, too, become soft when their interspacing falls under a critical size of a few nanometers, due to imperfections.
Scientists noted, “Very roughly speaking, nanocrystals are like patches of cloth and nanotwins are like strong but tiny threads in the cloth. Except they’re at the atomic scale. The new research combines both approaches to make what the scientists call a nanocrystalline-nanotwinned metal that has unique mechanical and physical properties.”
Sansoz explained, “That’s because the copper atoms, slightly smaller than the atoms of silver, move into defects in both the grain boundaries and the twin boundaries. This allowed the team—using computer simulations of atoms as a starting point and then moving into real metals with advanced instruments at the National Laboratories—to create the new super-strong form of silver.”
“The tiny copper impurities within the silver inhibit the defects from moving, but are such a small amount of metal—less than one percent of the total—that the rich electrical conductivity of silver is retained.”
“The copper atom impurities go along each interface and not in between. So they don’t disrupt the electrons that are propagating through.”
Fascinatingly, the metal overcomes the softening previously observed as grains. Also, the twin boundaries get excessively small—the so-called “Lobby Petch breakdown.” What’s more, it even surpasses the long-standing hypothetical Hall-Petch limit.
Sansoz said, “We’ve broken the world record, and the Hall-Petch limit too, not just once but several times in the course of this study, with very controlled experiments.”
“I hope the approach could be used to create super-strong and still-conductive silver that can be applied to many other metals. This is a new class of materials, and we’re just beginning to understand how they work.”
The team’s results were published on September 23 in the journal Nature Materials.
