Glass, an inorganic solid material that is usually transparent or translucent as well as hard, brittle, and impervious to the natural elements. Any crack, when split by bending, will cause the glass to break completely.
Lothar Wondraczek, with the University of Jena, has published a study in the journal Science outlining the history of scientists attempting to overcome the brittleness of glass.
Wondraczek noted, “scientists have been searching for ways to make the glass less brittle for as long as people have been making glass. The bendable glass would mean drinking glasses that survive a fall or smartphone screens that do not crack. In this new effort, the researchers say they have taken a step toward that goal.”
The primary composition of glass is silica (SiO2). Under suitable conditions, silicon combines with oxygen, and silica its produced. The main source of (natural source) silica is sand. Silica melts at about 1600°C. When it is heated at high temperature, it turns into glass.
In this new work, scientists used crystalline aluminum oxide instead of sand to make some tiny glass samples. To do so, the intense bursts of laser light were fired at a sample to turn it into a purple plasma. They then kept the material for cooling on a substrate.
The resulting material was sheets 60 nanometers thick and two micrometers wide. Also, it was transparent and extremely brittle than ordinary glass.
During the experiment, scientists showed that the glass could be bent or stretched.
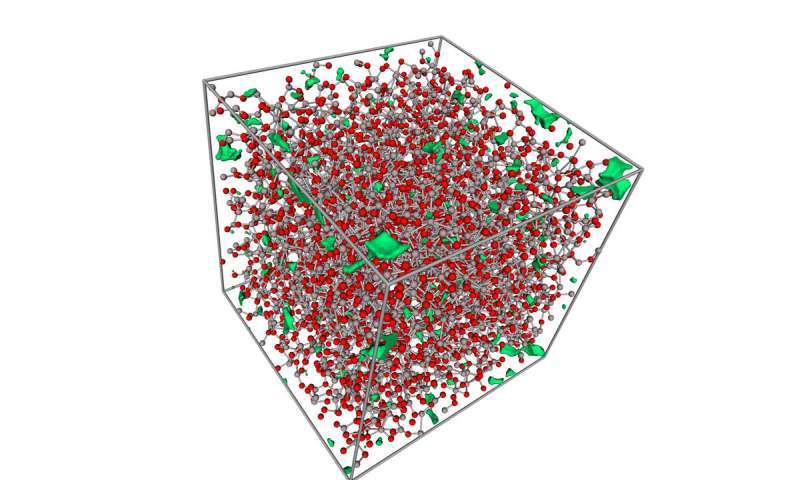
Scientists also observed their bendable glass using an electron microscope. They also created computer simulations of the material to better understand its properties.

The model showed that the glass had:
- A very tightly packed network of atoms that was defect-free, making it bendable.
- Its atoms were able to switch places when exposed to pressure.
However, further work is required before the bendable glass can be commercialized. It is not clear yet that the process can be used to make bigger sheets of glass, or if it is even amenable to manufacturing.