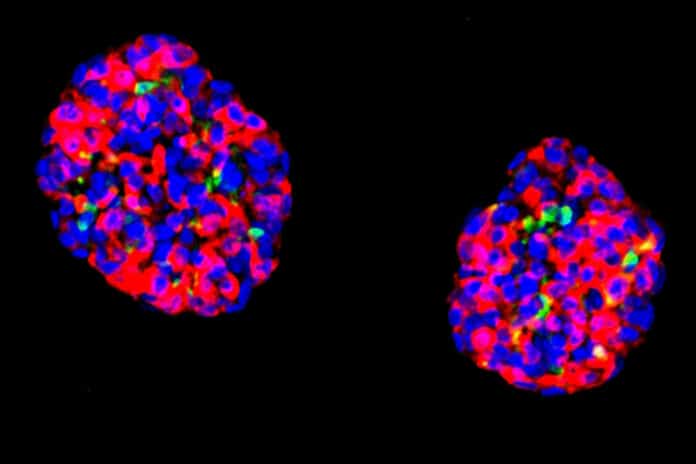
The ability to acquire gut stem cells via biopsy and have a significant proliferative capacity in culture make them an invaluable resource for autologous cell treatments. In the mouse gut, insulin-producing cells can be produced. Still, human gut tissues have not been able to produce an abundance or durability of insulin-secreting cells to assess their potential as a cell treatment for diabetes.
In a new study, scientists from Weill Cornell Medicine showed that stem cells from human stomach can be converted into insulin-secreting cells. Scientists demonstrated that they could obtain the stem cells from the human stomach and reprogram them directly—with strikingly high efficiency—into cells that closely resemble pancreatic insulin-secreting cells known as beta cells.
In experiments on a mouse model, this approach reversed disease signs. According to scientists, the study offers a promising approach, based on patient’s cells, for type 1 diabetes and severe type 2 diabetes.
With human cell transplants that operate like beta cells, autonomously sense blood sugar levels, and secrete insulin as required, biomedical researchers hope to restore beta-cell function more naturally. To avoid the issue of transplant rejection, such transplants should ideally be performed using the patient’s cells.
Study senior author Dr. Joe Zhou, a professor of regenerative medicine and a member of the Hartman Institute for Therapeutic Organ Regeneration at Weill Cornell Medicine, said, “The stomach makes its hormone-secreting cells, and stomach cells and pancreatic cells are adjacent in the embryonic stage of development, so in that sense, it isn’t completely surprising that gastric stem cells can be so readily transformed into beta-like insulin-secreting cells.”
The researchers transformed human stomach stem cells into beta-like cells, cultivated the cells in tiny clusters known as organoids, and discovered that these organ-like pieces of tissue swiftly developed glucose sensitivity and insulin release in response. The beta-like organoids performed essentially as real pancreatic beta cells would need when implanted into diabetic mice, secreting insulin in response to increased blood glucose and maintaining blood glucose levels. Additionally, the transplanted cells continued functioning for six months as the researchers observed them, indicating good durability.
Dr. Zhou said that he and his lab still need to optimize their method in various ways before it can be considered for clinical use. Necessary improvements include methods to increase the scale of beta-cell production for transplants to humans and modifications of the beta-like cells to make them less vulnerable to the type of immune attack that initially wipes out beta cells in type 1 diabetes patients.
Journal Reference:
- Huang, X., Gu, W., Zhang, J., et al. Stomach-derived human insulin-secreting organoids restore glucose homeostasis. Nat Cell Biol 25, 778–786 (2023). DOI: 10.1038/s41556-023-01130-y