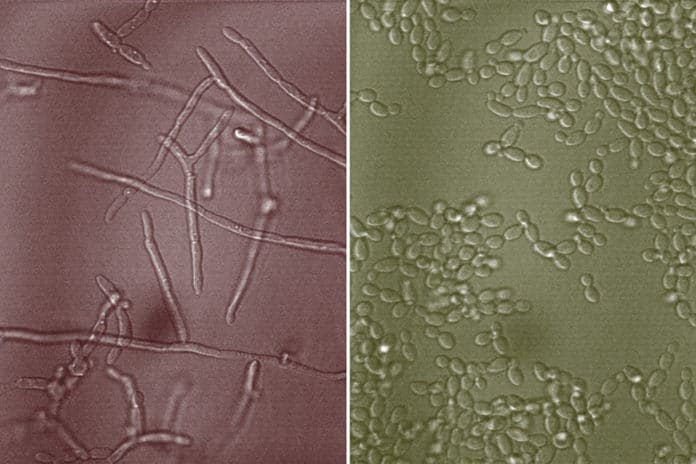
Mucins are huge gel-forming polymers found inside the mucus barrier that prevent Candida albicans from transitioning from yeast to hyphal, a significant virulence feature of this important human fungal disease. Despite their potential for therapeutic intervention, the molecular patterns in mucins that hinder filamentation remain unknown.
MIT scientists have now discovered mucus components that interact with Candida albicans and inhibit infection. These molecules, known as glycans, are a significant constituent of mucins, the gel-forming polymers that makeup mucus.
Mucins consist of several glycans, complex sugar molecules. According to research, glycans can be specialized to help tame specific pathogens such as Candida albicans, Pseudomonas aeruginosa, and Staphylococcus aureus.
Katharina Ribbeck, the Andrew and Erna Viterbi Professor at MIT, said, “The emerging picture is that mucus displays an extensive small-molecule library with lots of virulence inhibitors against all sorts of problematic pathogens, ready to be discovered and leveraged.”
Using these mucins could help scientists develop new antifungal treatments or make disease-causing fungus more vulnerable to existing drugs. There are few such medications on the market right now, and some harmful fungi have acquired resistance.
The previous study has suggested that mucins can prevent Candida albicans cells from switching from their round yeast form to multicellular filaments called hyphae, which is the harmful version of the microbe. Hyphae can secrete toxins that damage the immune system and the underlying tissue and are essential for biofilm formation, a hallmark of infection.
MIT graduate student Julie Takagi, the paper’s lead author, said, “Most Candida infections result from pathogenic biofilms, which are intrinsically resistant to the host immune system and antifungal therapeutics, posing significant clinical challenges for treatment.”
Mucus within yeast cells continues to grow and thrive but doesn’t become pathogenic. There must be something in mucus that has evolved over millions of years to keep pathogens in check.
In this study, scientists wanted to determine whether glycans could disarm Candida albicans on their own, detached from the mucin backbone, or if the entire mucin molecule is necessary.
For their study, scientists isolated glycans from the backbone and exposed them to Candida albicans. They found that these collections of glycans could prevent single-celled Candida from forming filaments. They may also inhibit adhesion and biofilm development and change the dynamics of Candida Albicans interactions with other microorganisms. Mucin glycans from human saliva and animal gastric and intestinal mucus were similar.
Isolating single glycans is difficult, hence scientists synthesized six different glycans that are most abundant on mucosal surfaces. They used them to test whether individual glycans can disarm Candida albicans.
Rachel Hevey, a research associate at the University of Basel, said, “Individual glycans are nearly impossible to isolate from mucus samples with current technologies. The only way to study the characteristics of individual glycans is to synthesize them, which involves extremely complicated and lengthy chemical procedures.”
After testing, they found that each of these glycans showed at least some ability to stop filamentation independently. Some were as potent as the collections of multiple glycans the researchers had previously tested.
According to a study on Candida gene expression, more than 500 genes are upregulated or downregulated in response to interactions with glycans. These genes included filament and biofilm formation genes and genes involved in the amino acid synthesis and other metabolic processes. Many of these genes appear to be regulated by the transcription factor NRG1, a master regulator triggered by glycans.
Ribbeck says, “The glycans seem to tap into physiological pathways and rewire those microbes. It’s a huge arsenal of molecules that promote host compatibility.”
Micheal Tiemeyer, a professor of biochemistry and molecular biology at the University of Georgia, said, “The analyses performed in this study also allowed the researchers to link specific mucin samples to the glycan structures found within them, which should allow them to explore further how those structures are correlated with microbial behaviors.”
“Using state-of-the-art glycomic methods, we have begun to define the richness of mucin glycan diversity comprehensively and to annotate that diversity into motifs that have functional implications for both host and microbe.”
By harnessing the variety of glycans, scientists could one day develop new treatments targeting different infectious diseases. As one example, glycans could be used to either halt a Candida infection or help sensitize it to existing antifungal drugs by breaking up the filaments they form in the pathogenic state.
Ribbeck says, “The glycans alone can potentially reverse an infection and convert Candida to a growth state that is less harmful to the body. They might also sensitize the microbes to antifungals because they individualize them, thereby making them more manageable by immune cells.”
Journal Reference:
- Takagi, J., Aoki, K., Turner, B.S., et al. Mucin O-glycans are natural inhibitors of Candida albicans pathogenicity. Nat Chem Biol (2022). DOI: 10.1038/s41589-022-01035-1