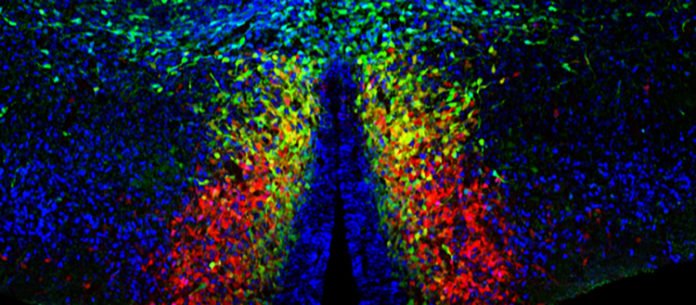
The cerebral cortex comprises a substantial assorted variety of neurons, each showing particular qualities regarding atomic, morphological and utilitarian highlights. Yet, where are these neurons conceived? How would they build up their particular properties? We don’t presently have clear responses to these inquiries, to a great extent as a result of methodological confinements.
Now, UNIGE scientists have discovered a unique molecular factor allowing them to track, from birth to maturity, a homogeneous class of neurons called the neurogliaform cells. Their study reveals that the beginning and the improvement of these cells, opening new open doors for the comprehension of neuronal specificities and working of the cerebral cortex.
In order to answer the questions, scientists proposed two different types of models. The first consideration that the different classes of neurons are born with the same identity, and only later specialize depending on their environment. The second model states that an initial genetic diversity exists, which, as early as birth, drives neurons to specify features of a given class.
Through these models, scientists can successfully locate precisely the genesis of a specific category of inhibitory neurons and to track down its path towards the cerebral cortex.
The team led by Alexandre Dayer, Professor in the Departments of Psychiatry and Basic Neurosciences at UNIGE’s Faculty of Medicine said, “We observed and tested a number of different transcription factors responsible for gene regulation. We found that the gene HMX3 is expressed very locally in a region known to generate interneurons. This could be the initial genetic fingerprint of a precise class!”
“We found that the gene HMX3 can actually be used to follow the developmental trajectory of a unique subtype of interneuron: the neurogliaform cells.”
This discovery is an important step forward in understanding the occurrence of neuronal diversity. Our results show that early genetic factors are responsible for the specialization of neurogliaform cells, and this concept is probably fitting with what happens to the other twenty interneuron subtypes present in the cerebral cortex.”
Mathieu Niquille, a researcher in the Department of Psychiatry at UNIGE’s Faculty of Medicine said, “Thanks to a genetically-modified mouse that makes the HMX3 lineage visible, we could uncover the origins of neurogliaform cells. We have suggested that this subtype of interneuron is defined by the expression of this transcription factor.”
“Not only this group of interneuron has its origins in a zone called the preoptic area, located far from the cerebral cortex, but it is specialized already at birth. This gives credit to the second model proposed by the scientific community.”
Dayer said, “This research also opens up new horizons for a better understanding of psychiatric diseases, such as autism and schizophrenia, which are particularly affected by alterations of the balance between inhibitory and excitatory neurons at early stages of development.”
The results are published in the journal e-life.