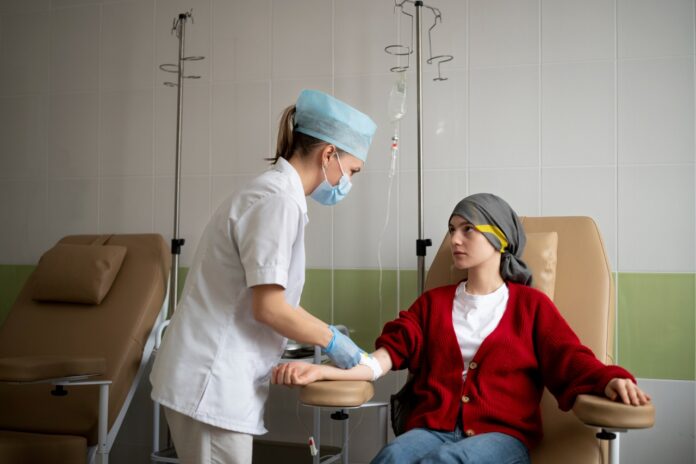
Chemotherapy is a cancer treatment that uses drugs to destroy cancer cells in the body. However, one of the biggest challenges of chemotherapy is getting the drugs to reach the cancer cells in the brain. This is because the blood-brain barrier, which separates the blood vessels in the brain from the rest of the body, can prevent chemotherapy drugs from reaching the brain.
Northwestern Medicine scientists have successfully used a skull-implantable ultrasound device to permeate the blood-brain barrier and deliver chemotherapy to patients with glioblastoma, a deadly brain cancer. The four-minute procedure, performed with the patient awake, led to a four- to six-fold increase in drug concentrations in the human brain.
The treatment was safe and well-tolerated, with some patients receiving up to six treatment cycles. This breakthrough could pave the way for more effective treatments for brain cancer and other neurological diseases.
“This is potentially a huge advance for glioblastoma patients.” said lead investigator Dr. Adam Sonabend, an associate professor of neurological surgery at Northwestern University Feinberg School of Medicine and a Northwestern Medicine neurosurgeon.
Temozolomide, the current chemotherapy used for glioblastoma, does cross the blood-brain barrier but is a weak drug, Sonabend said.
CED involves using a catheter to deliver drugs directly into the brain. The catheter is inserted through a small hole in the skull and placed into the tumor. The drug is then infused into the tumor slowly and steadily, allowing it to diffuse throughout the tumor and kill the cancer cells.
The study involved ten patients with recurrent glioblastoma, an aggressive type of brain cancer. The patients were treated with a single dose of doxorubicin delivered using CED. The researchers found that the drug could reach the tumor without serious side effects.
This is an essential breakthrough in cancer treatment, as it could lead to more effective treatments for brain cancer. The ability to deliver chemotherapy drugs directly to the brain could also be useful in treating other neurological diseases, such as Parkinson’s disease and Alzheimer’s disease.
However, the researchers caution that more research is needed to determine this approach’s long-term safety and efficacy. They also note that the technique is currently only available in specialized medical centers and may only be suitable for some patients.
Researchers conducted a phase 1 clinical trial in adults with recurrent glioblastoma using a nine-emitter ultrasound device implanted into a skull window after tumor resection. Patients received LIPU-MB with intravenous albumin-bound paclitaxel infusion every three weeks for up to six cycles.
Six dose levels of albumin-bound paclitaxel were evaluated to determine dose-limiting toxicity occurring during the first cycle of sonication and chemotherapy. Safety was assessed in all treated patients, and blood-brain barrier opening was investigated using MRI. Pharmacokinetic analyses were also done in a subgroup of patients. A phase 2 trial is currently open for accrual.
In the phase 1 clinical trial, researchers successfully used an implantable ultrasound device to repeatedly open the blood-brain barrier and deliver albumin-bound paclitaxel to patients with recurrent glioblastoma.
Blood-brain barrier opening was investigated using MRI and pharmacokinetic analyses in a subgroup of patients. These promising results suggest that this approach could effectively treat glioblastoma.
In conclusion, the phase 1 clinical trial demonstrated the feasibility and safety of using an implantable ultrasound device to repeatedly open the blood-brain barrier and deliver albumin-bound paclitaxel to patients with recurrent glioblastoma. This approach showed promising results, and a phase 2 trial is currently open for further evaluation.
This breakthrough could pave the way for more effective treatments for brain cancer and other neurological diseases.
Journal Reference:
- Adam M Sonabend, Andrew Gould, Christina Amidei, et al. Repeated blood–brain barrier opening with an implantable ultrasound device to deliver albumin-bound paclitaxel in patients with recurrent glioblastoma: a phase 1 trial. The Lancet Oncology DOI:10.1016/S1470-2045(23)00112-2