Microfluidics refers to small-scale machines that handle fluids and assist in chemical reactions. They have various applications, including detecting diseases in small blood or fluid samples. For instance, at-home COVID-19 test kits use a basic form of microfluidics.
However, certain microfluidic applications involve chemical reactions that require specific temperatures. More advanced microfluidic devices, usually produced in clean rooms, incorporate heating elements made from gold or platinum. The fabrication process for these elements is complex, expensive, and challenging to scale up. This complexity and cost have hindered the widespread adoption of more sophisticated microfluidic technologies.
MIT scientists have used 3D printing to produce self-heating microfluidic devices. They demonstrated a technique to create cheap yet accurate tools to detect various diseases.
The MIT team took a different approach by utilizing multimaterial 3D printing to craft self-heating microfluidic devices with integrated heating elements, all through a single and cost-effective manufacturing process. They designed devices that can warm up fluids to precise temperatures as these fluids move through microscopic channels within the miniature apparatus.
Their method is flexible; engineers can tailor it to create a microfluidic device that heats fluids to a specific temperature or follows a particular heating pattern within a designated section of the device. This adaptability makes the technique versatile for various applications and allows for customizing microfluidic devices based on specific needs.
This innovative fabrication process employs multi-material extrusion 3D printing, a technique where various materials are extruded through multiple nozzles of the printer to construct a device layer by layer. The key feature of this process is its monolithic nature, meaning the entire device can be manufactured in a single step on the 3D printer, eliminating the need for any post-assembly steps.
In creating self-heating microfluidic devices, the researchers utilized two materials: a biodegradable polymer called polylactic acid (PLA), commonly used in 3D printing, and a modified version of PLA. The modified PLA contains copper nanoparticles mixed into the polymer, transforming this insulating material into an electrical conductor. When electrical current is applied to a resistor made from this copper-doped PLA, it dissipates energy as heat, providing a reliable and integrated heating element for the microfluidic device.
Velásquez-García explains, “It is amazing when you think about it because the PLA material is a dielectric, but when you put in these nanoparticle impurities, it completely changes the physical properties. We don’t fully understand this yet, but it happens and is repeatable.”
Using a multi-material 3D printer, the scientists first created a heating resistor using copper-doped PLA. In a single printing step, they then produce the microfluidic device directly on top with microscopic channels for fluid flow. This is possible because both components are made from the same base material, ensuring compatibility and similar printing temperatures.
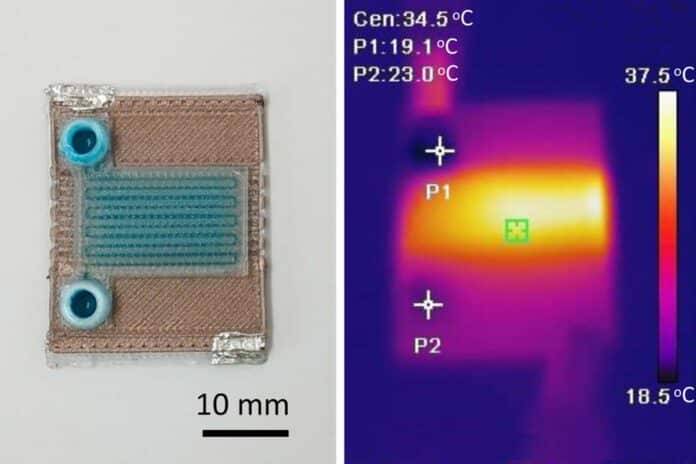
The heat generated by the resistor transfers to the fluid flowing through the channels in the microfluidic device.
In addition to the resistor and microfluidic device, the scientists employ the 3D printer to add a thin, continuous layer of PLA sandwiched between them. The challenge lies in manufacturing this layer with precision—it must be light enough for efficient heat transfer from the resistor to the microfluidic device yet not so thin that fluid could potentially leak into the resistor. This intricate process ensures the functionality and integrity of the self-heating microfluidic device.
The final device is roughly the size of a U.S. quarter and can be manufactured in just a few minutes. It features channels approximately 500 micrometers wide and 400 micrometers tall that weave through the microfluidic structure to transport fluids and enable chemical reactions.
A crucial aspect of this device is that the PLA material used is translucent, allowing for the visibility of fluids inside the device. This transparency is valuable because many processes depend on visualization or the use of light to understand and observe what is occurring during chemical reactions within the microfluidic system.
Using this one-step manufacturing process, scientists generated a prototype that could heat fluid by 4 degrees Celsius as it flowed between the input and the output. Thanks to this adaptable technology, they can create devices that heat fluids in specific patterns or along particular gradients.
Scientists noted, “You can use these two materials to create chemical reactors that do exactly what you want. We can set up a particular heating profile while still having all the capabilities of the microfluidic.”
While the developed microfluidic device has notable advantages, a limitation arises from PLA starting to degrade at temperatures around 50 degrees Celsius. This becomes restrictive for specific chemical reactions, like those employed in polymerase chain reaction (PCR) tests, which often require temperatures of 90 degrees Celsius or higher. To address this, incorporating a third material into the system for temperature sensing would be necessary to control the device’s temperature precisely.
In the future, scientists are looking forward to overcoming these limitations. Additionally, they are interested in integrating magnets directly into the microfluidic device for performing chemical reactions that involve sorting or aligning particles within the device.
Scientists noted, “If we can understand the mechanism related to the electrical conductivity of PLA, that would greatly enhance the capability of these devices, but it is going to be a lot harder to solve than some other engineering problems.”
Norihisa Miki, a professor of mechanical engineering at Keio University in Tokyo, said, “In Japanese culture, it’s often said that beauty lies in simplicity. The work of Cañada and Velasquez-Garcia echoes this sentiment. Their proposed monolithically 3D-printed microfluidic systems embody simplicity and beauty, offering a wide array of potential derivations and applications we foresee in the future.”
Niclas Roxhed, an associate professor at Sweden’s KTH Royal Institute of Technology, said, “Being able to directly print microfluidic chips with fluidic channels and electrical features at the same time opens up very exiting applications when processing biological samples, such as to amplify biomarkers or to actuate and mix liquids. Also, due to the fact that PLA degrades over time, one can even think of implantable applications where the chips dissolve and resorb over time.”
The research will be presented at the PowerMEMS Conference this month.