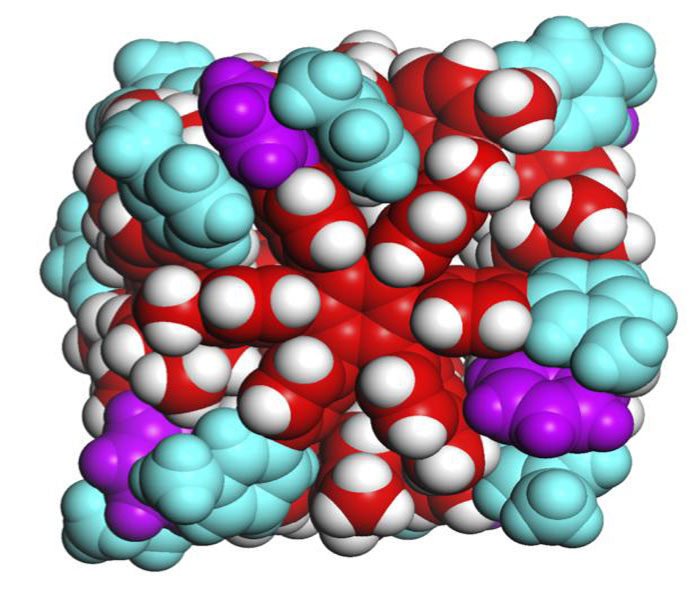
Scientists at the University of Tokyo have identified a weak molecular force that holds together a modest, self-assembling box with great conceivable outcomes. The examination exhibits a useful utilization of a force basic in biological systems and advances the quest for synthetic chemical life.
The formation of DNA and proteins are organic models of self-assembly, however, the powers or process controlling how these natural molecules come together stay unclear. Examinations by Hiraoka’s group add to a chemical understanding of how natural molecules may self-assemble and uncover procedures for copying those procedures later on.
Professor Shuichi Hiraoka, leader of the laboratory at the University of Tokyo Graduate School of Arts and Sciences said, “I want to understand self-assembly systems, which are essential for life. Building artificial self-assembling cubes helps us understand how biological systems function.
CREDIT
Image by Shuichi Hiraoka CC-BY-ND.
Scientists identified the forces holding together the sides of their small boxes as van der Waals powers, for the most part dispersion forces. These powers are weak attractions between particles made when electrons briefly assemble together on one side of an atom. Geckos can stroll up walls to a limited extent because of van der Waals forces.
Each side of the cube is framed from one atom that is 2 nanometers in diameter and formed like a six-pointed snowflake. Each side is around one-four-thousandth the extent of a human platelet.
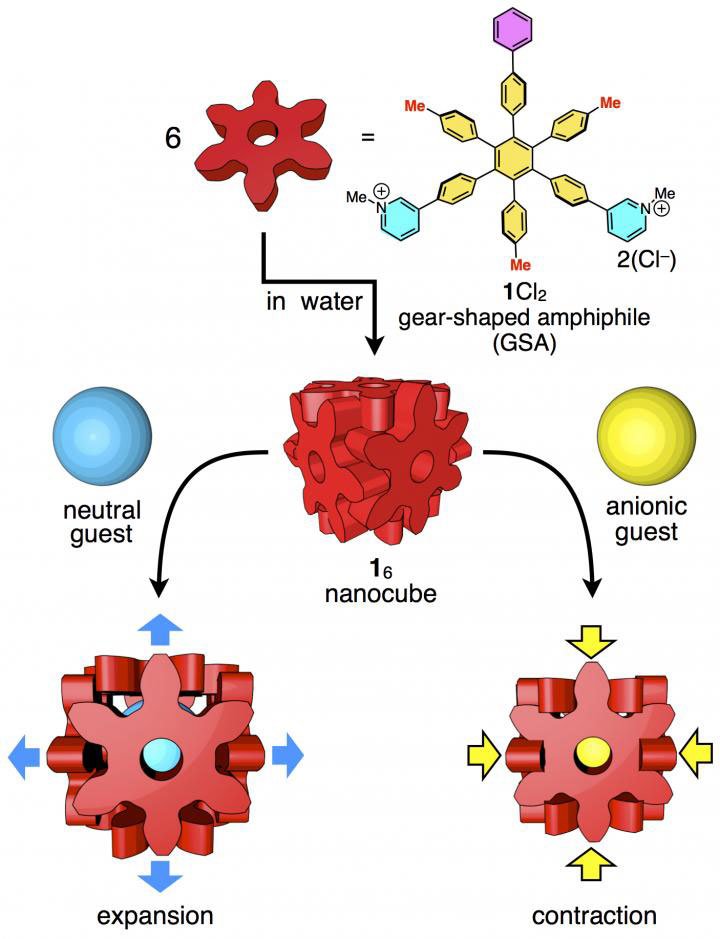
The weak forces holding the sides of the block together make the crate marginally adaptable, so it changes with the best suit accommodate particles dependent on their size, shape, and nuclear charge. The box can lump to hold long or long contents and contract to take out additional space while facilitating visitor atoms with a negative charge(s).
Scientists developed the tiny box out of molecules of hexaphenylbenzene. The individual molecules exist as a dry, white powder. When mixed with water, the molecules spontaneously self-assemble into cubes.
The six points of the snowflake-shaped hexaphenylbenzene molecules lock together when they assemble into a cube. Researchers describe the design of this molecular box as resembling the Japanese wood joining technique called hozo, where pieces of wood are held together without adhesives or hinges, using only intricate interlocking designs.
Hiraoka said, “In solution, the six molecules come together so quickly that we cannot observe how they form cubes. The exact process of self-assembly remains a mystery.”
The study is published in the journal Nature Communications.