Fast charging rate and large energy storage are becoming key elements for the development of next-generation batteries, especially for high-performance electric vehicles. Developing electrodes with high volumetric and gravimetric capacity that could be operated at a high rate is the most challenging part of this process.
Now, scientists at the UNIST, in collaboration with Professor Soojin Park and his research team at Pohang University of Science and Technology (POSTECH) have come up with a new way that could potentially lead to the development of next-generation batteries. They have developed new silicon-based coral-like anode materials to replace conventional graphite.
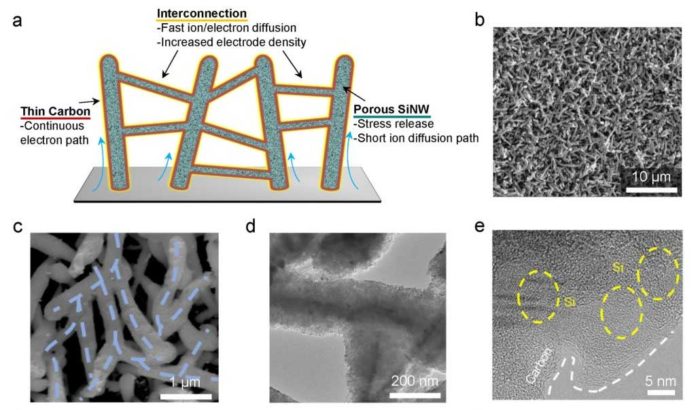
The benefit of using silicon-silicon as the anode material that it delivers the highest theoretical capacity as a lithium-ion battery anode. Using the material, scientists reported a binder-free electrode that interconnects carbon-sheathed porous silicon nanowires into a coral-like network and shows fast charging performance coupled to high energy and power densities when integrated into a full cell with a high areal capacity loading.
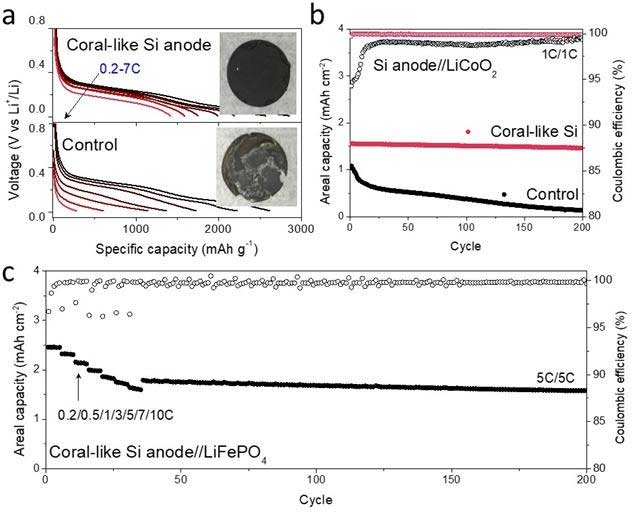
When scientists evaluated the battery performance of this new material, they found that the material can perform well even under ultrafast charging for test anodes in both half cell (pairing with a Li metal anode) and full cell (pairing with a cathode that closely mimics real commercial cathodes) as lithium-ion batteries (LIBs). The full batteries showed promising volumetric energy and power densities that might meet new expectations for fast-charging LIBs.
Co-first author Dr. Bin Wang said, “Several aspects were considered when designing this material: First, an interconnected three-dimensional network of silicon nanowires that improves electron transport in the entire electrode was targeted; Second, to promote ion transport, we combined the macro-pores between the silicon nanowires and the nano-pores inside the nanowires to provide channels for fast ion transport;”
“Finally, a conformal and thin coating of layers of carbon was achieved that reduced the resistance in the system, and also provided a barrier to stop this undesirable thickening of the SEI and to accommodate the volume change of the silicon nanostructures.”
“Through the collaboration with experts in China and Korea, the network of interconnected carbon-coated porous silicon nanowires as a binder-free anode in LIBs was achieved and tested in real battery analogs.”
Professor Soojin Park at POSTECH and co-first author Dr. Jaegeon Ryu from Prof. Soojin Park’s group thoroughly tested this new material as an anode in test cell LIBs. It was first assembled into a ‘half-cell’ to check its intrinsic properties. “This material exhibited excellent high-rate performance. At a 7 C-rate, it delivered a reversible capacity of 1275 mAh g-1 after 500 cycles, and its Coulombic efficiency per cycle reached 99.5% at the second cycle and the average Coulombic efficiency was 99.9% for further cycles. This high performance is closely related to the highly interconnected porous structure directly grown onto the current collectors and the conductive carbon sheath coating.”
The highest volumetric energy density from pairing with a LiCoO2 cathode was 1621 Wh L-1 and the power density from pairing with a LiFePO4 cathode reached 7762 W L-1.
Professor Park noted “The results are competitive in the field of silicon-based LIBs, among the best results from the previous silicon-based anodes and offers promise for meeting fast-charging requirements. When it comes to the future developments, if cathode materials are further developed with higher capacity and faster Li-ion transfer kinetics, the overall battery specifications can be further improved to advance the realization of Si-based anodes for fast-charging capability with high volumetric energy density.”
This research has been supported by the Institute for Basic Science, the Center for Advanced Soft-Electronics, and the National Natural Science Foundation of China. Professor Linjie Zhi and Professor Xianglong Li at the National Center for Nanoscience and Technology (NCNST) in the Chinese Academy of Sciences, and Professor Didier Pribat in the Sungkyunkwan University also partook in this study.
The study was published in the journal ACS Nano.