High entropy alloys (HEAs) are a group of metals that includes a subset of CrCoNi. HEAs are constructed of an equal mixture of each constituent element, unlike all current alloys, which all have a high proportion of one element and smaller additions of other elements. Some of these materials appear to possess an incredibly high mix of strength and ductility under stress, which combined constitute what is referred to as “toughness,” thanks to these balanced atomic compositions.
Since they were created roughly 20 years ago, HEAs have attracted much scientific attention. However, until recently, the equipment needed to test the materials to the absolute limit was not readily available.
In a new study, scientists reported about the toughest material on Earth. Scientists from Lawrence Berkeley National Laboratory (Berkeley Lab) and Oak Ridge National Laboratory have measured the highest toughness ever recorded of any material while investigating a metallic alloy made of chromium, cobalt, and nickel (CrCoNi). The metal is not only incredibly strong (meaning it resists permanent deformation) and exceedingly ductile (which, in materials science, means highly pliable), but it also becomes more ductile and strong as it cools. Contrary to the majority of other materials in use, this.
Project co-lead Easo George, the Governor’s Chair for Advanced Alloy Theory and Development at ORNL and the University of Tennessee, said, “When you design structural materials, you want them to be strong but also ductile resistant to fracture. Typically, it’s a compromise between these properties. But this material is both, and instead of becoming brittle at low temperatures, it gets tougher.”
Research co-leader Robert Ritchie, a senior faculty scientist in Berkeley Lab’s Materials Sciences Division and the Chua Professor of Engineering at UC Berkeley, said, “The toughness of this material near liquid helium temperatures (20 kelvin, -424 Fahrenheit) is as high as 500 megapascals square root meters. In the same units, the toughness of a piece of silicon is one, the aluminum airframe in passenger airplanes is about 35, and the toughness of some of the best steels is around 100. So, 500, it’s a staggering number.”
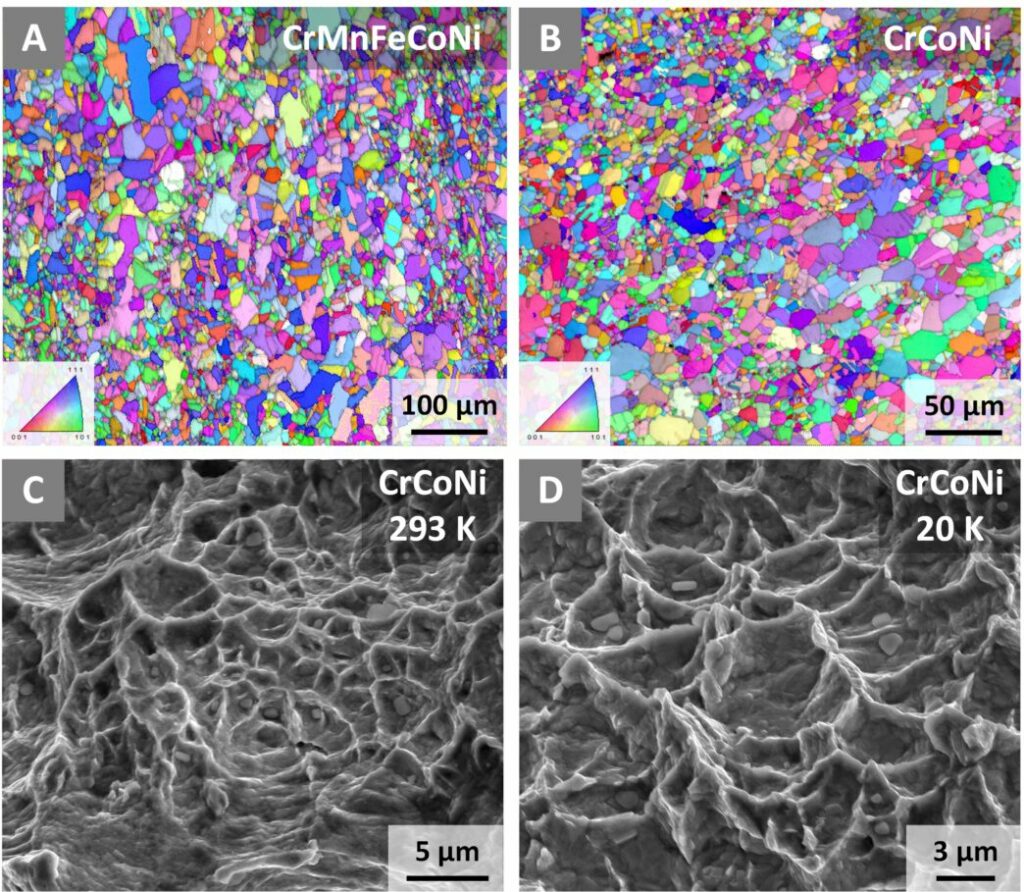
Scientists started experimenting with CrCoNi and another alloy containing manganese and iron (CrMnFeCoNi) nearly a decade ago. They created samples of the alloys, lowered the materials to liquid nitrogen temperatures (around 77 kelvin, or -321 F), and discovered impressive strength and toughness.
They wanted to conduct tests at liquid helium temperature ranges to confirm their findings immediately. Still, it took the team the next ten years to find facilities that would allow them to do so, as well as team members who had the necessary analytical skills and training to study the material’s behavior at the atomic level.
Scientists used neutron diffraction, electron backscatter diffraction, and transmission electron microscopy to observe the lattice structures of CrCoNi samples fractured at room temperature and 20 K. The images and atomic maps generated revealed that the alloy’s toughness is due to a trio of dislocation obstacles that come into effect in a particular order when force is applied to the material.
Areas of the crystal that are on parallel planes first slide away from one another as a result of sliding dislocations. This movement displaces layers of unit cells, and as a result, a form of obstruction is created in the direction perpendicular to the slipping movement. When the metal is subjected to further pressure, a process known as nanotwinned occurs in which portions of the lattice form a mirrored symmetry with a boundary in the middle. The CrCoNi atoms move from a face-centered cubic crystal to another arrangement known as tight hexagonal packing if forces continue to operate on the metal. This transformation occurs if forces continue to act on the metal.
Ritchie said, “This sequence of atomic interactions ensures that the metal keeps flowing but also meets new resistance from obstacles far past the point that most materials snap from the strain. So as you are pulling it, the first mechanism starts, and then the second one starts, and then the third one starts, and then the fourth.”
“Now, many people will say, well, we’ve seen nanotwinning in regular materials, we’ve seen a slip in regular materials. That’s true. There’s nothing new about that, but it’s a fact they all occur in this magical sequence that gives us these tremendous properties.”
“The findings, taken with other recent work on HEAs, may force the materials science community to reconsider long-held notions about how physical characteristics give rise to performance. It’s amusing because metallurgists say that the structure of a material defines its properties, but the structure of the NiCoCr is the simplest you can imagine – it’s just grains.”
Co-author Andrew Minor, director of the National Center of Electron Microscopy facility of the Molecular Foundry at Berkeley Lab and Professor of Materials Science and Engineering at UC Berkeley, said, “However, when you deform it, the structure becomes very complicated, and this shift helps explain its exceptional resistance to fracture. We were able to visualize this unexpected transformation due to the development of fast electron detectors in our electron microscopes, which allow us to discern between different types of crystals and quantify the defects inside them at the resolution of a single nanometer – the width of just a few atoms – which as it turns out, is about the size of the defects in deformed NiCoCr structure.”
Journal Reference:
- Dong Liu et al. Exceptional fracture toughness of CrCoNi-based medium- and high-entropy alloys at 20 kelvin. Science. DOI: 10.1126/science.abp8070
