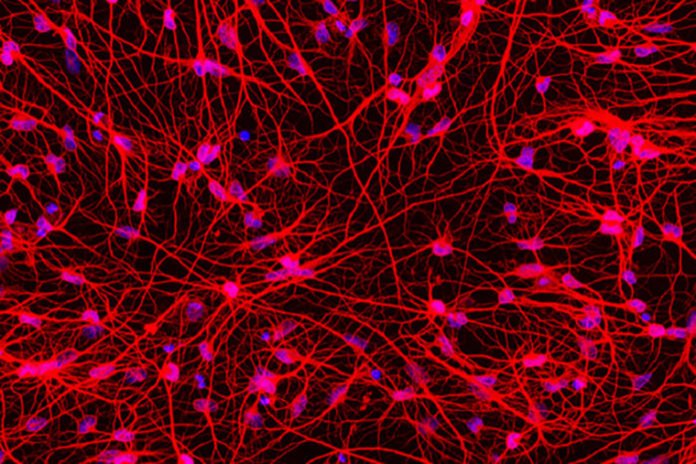
According to the researchers at Washington University School of Medicine in St. Louis, acquired a mass of neurons when they transformed skin cells from patients with Huntington’s disease into the kind of brain cell affected by the disorder can serve as a novel tool to study the degenerative and fatal neurological condition in due course.
A recently published study discovered that the patients’ nerve cells transformed outright from patients’ skin cells evinced “symptoms” of the disorder incorporating DNA detriment, dysfunctional mitochondria, and cell demise.
Further to rectify malfunctioning genes in these reprogrammed neurons avert the cell demise have attributes of Huntington’s disease, an inherited genetic disorder that causes cognitive fall and involuntary muscle movements.
Andrew S. Yoo, an assistant professor of developmental biology, said, “This is a powerful tool to investigate the reasons why particular brain cells with the disease-associated mutation become sick over time and eventually die.”
“In theory, we could model progression of the disease by reprogramming skin cells from patients at a range of ages, including before symptoms begin. And if there are drugs or compounds that may help these patients, we can test them first in this system.”
Huntington’s disease and other inherited brain conditions are challenging to study since it is arduous to receive samples of neurons from living patients. Succeeding to this the most prominent thing they discovered in various ways to transform skin cells into brain cells. However, skin cells are convenient to gather from patients and share the same genetic blueprint and disease-causing mutations as brain cells.
The researchers, accompanying Matheus Victor, a postdoctoral research associate, set out to produce neurons that would mimic those of adult patients in order to model the onset and progression of Huntington’s disease. They achieved this objective with outright conversion method they developed.
This particular process permits skin cells to bypass the stem cell stage as they are being reprogrammed into neurons. Going through a stem cell stage resets the development clock to an embryonic-like state, wiping out the age-associated impacts of the disorder.
According to Yoo and his team, the directly reprogrammed neurons retain their age, along with the problems associated with adult-onset Huntington’s disease.
In this outright reprogramming, the researchers revealed the adult skin cells to a particular blend of signaling particles discovered in the past research which would convert healthy skin cells outright to a kind of brain cell called medium spiny neurons, without intermediate steps along the way. Further, this signaling cocktail repackages the DNA, folding up the instructions for skin cells and unfurling the instructions for neurons.
In this process, the resulting reprogrammed medium spiny neurons retain the patient’s chronological age, along with age-associated symptoms of the disorder.
Although other neuronal cell types are affected, medium spiny neurons bear the brunt of the detriment caused by Huntington’s disease. The genetic error that causes the disease leads a key protein, Huntingtin, to be misshapen and not function properly.
Post this process, result demonstrate the malfunctioning protein builds up and by some set of events that remains unknown, in due course kills the cell.
Yoo said, since the neuronal demise can be recapitulated in directly reprogrammed patient neurons, the novel technique offers a way to study the details of how potential therapies including medications that are currently being tested in clinical trials could rescue medium spiny neurons from demise.
The team of researchers comprehending the inside information of how the disease progresses to cause cell demise discovered another prominent protein. This particular protein eminent normal medium spiny neurons named as SP9, but previously had not associated with Huntington’s disease.
Lastly, in the study, the researchers discovered significantly less SP9 in the transformed medium spiny neurons from Huntington’s patients in which they gave back SP9 to the diseased cells and found that returning this protein to the diseased neurons reduced cell demise to levels analogous to healthy control cells.
Yoo stated, “We want to understand what drives the disease progression over time.”
“Most neurodegenerative disorders worsen over time, so this method of modeling potentially could be applied in other conditions. This technique lets us capture characteristics of the disease at distinct moments in its progression. That’s important to understand what is happening and finding ways to stop it.”